
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
13th Edition
ISBN: 9780137444298
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.4, Problem 12.36PP
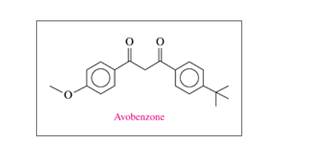
Avobenzone is a common ingredient in sunscreen. Its structural formula is shown.
a. What
b. What is the molecular formula and molar mass of avobenzone?
c. If a bottle of sunscreen containing 236 mL has 3.0% (m/v) avobenzone, how many grams of avobenzone are present?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
molar mass =
228.2
g/mol
molecular formula of oxybenzone is C14H12O3
If a bottle of sunscreen containing 161 mL has 6.0 % (m/v) oxybenzone, how many grams of oxybenzone are present?
12.36 Avobenzone is a common ingredient in sunscreen. Its
structural formula is shown.
a. What functional groups are in avobenzone?
b. What is the molecular formula and molar mass of
avobenzone?
c. If a bottle of sunscreen containing 236 mL has 3.0% (m/v)
avobenzone, how many grams of avobenzone are present?
Avobenzone
0.1% w/v = ? mg/100 ml? Only include the number and not any units
2) 0.1% = ? ug/ml? Only include the number and not any units
3) What is the concentration of your stock in ug/ml? Only include the number and not any units
4)The molecular weight of tartrazine is 534.4 g/mole. What is the concentration of your stock solution in mM? Be sure to give your answer in milliMolar to TWO decimal places.
Chapter 12 Solutions
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Prob. 12.3PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Give the common name for each of the following: a....Ch. 12.1 - Prob. 12.6PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Draw the condensed structural formula, or...Ch. 12.2 - Classify each of the following alcohols as primary...Ch. 12.2 - Classify each of the following alcohols as primary...
Ch. 12.2 - Prob. 12.11PPCh. 12.2 - Prob. 12.12PPCh. 12.2 - Give an explanation for each of the following...Ch. 12.2 - Give an explanation for each of the following...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Prob. 12.27PPCh. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Prob. 12.33PPCh. 12.4 - Prob. 12.34PPCh. 12.4 - Oxybenzone is an effective sunscreen whose...Ch. 12.4 - Avobenzone is a common ingredient in sunscreen....Ch. 12 - Prob. 12.37UTCCh. 12 - The compound frambinone has the taste of...Ch. 12 - A compound called resveratrol is an antioxidant,...Ch. 12 - A compound called cinnamaldehyde is found in...Ch. 12 - Prob. 12.41UTCCh. 12 - Prob. 12.42UTCCh. 12 - Prob. 12.43APPCh. 12 - Classify each of the following alcohols as primary...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Prob. 12.63CPCh. 12 - Draw the condensed structural formulas and give...Ch. 12 - A compound with the formula C4H8O is synthesized...Ch. 12 - A compound with the formula C5H10O oxidizes to...Ch. 12 - Compound A is a primary alcohol whose formula is...Ch. 12 - Compound X is a secondary alcohol whose formula is...Ch. 12 - Prob. 21CICh. 12 - Prob. 22CICh. 12 - Prob. 23CICh. 12 - Prob. 24CICh. 12 - Prob. 25CICh. 12 - lonone is a compound that gives violets their...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. IF0.5 mol of salicylic acid (CHOs) is needed for a reaction with acetic anhydride, how many L of a 5 (w/v)% solution should be used?arrow_forwardIf a bottle of sunscreen containing 187 mL has 4.0 % (m/v) avobenzone, how many grams of avobenzone are present?arrow_forward1) 0.1% w/v = ? mg/100 ml? Only include the number and not any units 2) 0.1% = ? ug/ml? Only include the number and not any units 3) What is the concentration of your stock in ug/ml? Only include the number and not any units 4)The molecular weight of tartrazine is 534.4 g/mole. What is the concentration of your stock solution in mM? Be sure to give your answer in milliMolar to TWO decimal places. 5)What dilution of your stock do you need to get the first standard (100 ug/ml)? Express as 1:x, where x is the fold dilution needed. 6)What weight of tartrazine (in mg) would you need to make up 250 mL of a 2.5 mM solution (MW of tartrazine is 534.4 g/mole)? Give your answer to the nearest whole number. write the number only, not the unitsarrow_forward
- 1) 0.1% w/v = ? mg/100 ml? Only include the number and not any units 2) 0.1% = ? ug/ml? Only include the number and not any units 3) What is the concentration of your stock in ug/ml? Only include the number and not any units 4)The molecular weight of tartrazine is 534.4 g/mole. What is the concentration of your stock solution in mM? Be sure to give your answer in milliMolar to TWO decimal places. 5)What dilution of your stock do you need to get the first standard (100 ug/ml)? Express as 1:x, where x is the fold dilution needed.arrow_forwardHow many milliliters of a syrup containing 85% w/v of sucrose should be mixed with 150 mL of a syrup containing 60% w/v of sucrose to make a syrup containing 80% w/v of sucrose?arrow_forwardWhat volumes of Tequila (40% ABV) and fruit juice will be used to make 350mL of Margarita with 10% ABV?arrow_forward
- A pharmacist fills a prescription for 30 g of a 0.1% w/w hydrocortisone cream by combining a 1% w/w hydrocortisone cream and a cream base. H ow many grams of each were used?arrow_forwardHow many milliliters of a syrup containing 85% w/v of sucrose should be mixed with 150 mL of a syrup containing 60% w/v of sucrose to make a syrup containing 80% w/v of sucrose? A) 600 mL B) 400 mL C) 550 mL D) 625 mLarrow_forwardHow many grams of hexynyl lithium should be produced? 1-hexyne (0.22 mL) + LiHMDS (1.0 mL of 1.0 M solution in THF) = hexynyl lithiumarrow_forward
- What is the % (v/v) concentration of a solution formed by mixing 5.06 mL of 2-propanol in enough water to make a solution with a volume of 36.9 mL? O 15.9% (v/v) O 0.159% (v/v) O 84.1% (v/v) O 13.7% (v/v)arrow_forwardWhat is the new concentration in % (w/v) of a KOH solution made by diluting 110 mL of a 6 % (w/v) KOH solution to 330 mL? A. 2 % B. 1% C. 6% D. 12 % E. 18 %arrow_forwardHow many milliliters of a 0.1% (w/v) thimerosal topical solution should be used to prepare one-half quart of a 1:5000 solution for irrigation? 473 94.6 O 47.3 O 946arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY