
ORGANIC CHEMISTRY-EBOOK>I<
9th Edition
ISBN: 9781305084414
Author: McMurry
Publisher: INTER CENG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.8, Problem 10P
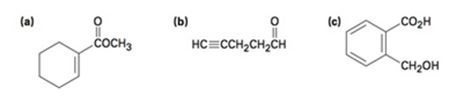
Where might the following compounds have IR absorptions?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Explain the Effect of Resonance on IR Absorptions ?
Which compound has the infrared spectrum shown?
HC
Citral
Citronellal
MICRONS
9
10
11
12
13
14 15
16
19
25
10025
90
80
70
60
50
40
30
20
1720 cm
÷1
10
4000
3600
3200
2800
2400
2000
1800
1600
1400
1200
1000
800
600
400
WAVENUMBERS (CM-")
What is the compound of this IR Spectrum?
Chapter 12 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
Ch. 12.2 - Prob. 1PCh. 12.2 - Two mass spectra are shown in FIGURE 12-8. One...Ch. 12.3 - What are the masses of the charged fragments...Ch. 12.3 - Prob. 4PCh. 12.5 - Prob. 5PCh. 12.5 - Prob. 6PCh. 12.7 - What functional groups might the following...Ch. 12.7 - How might you use IR spectroscopy to distinguish...Ch. 12.8 - Prob. 9PCh. 12.8 - Where might the following compounds have IR...
Ch. 12.8 - Where might the following compound have IR...Ch. 12.SE - Prob. 12VCCh. 12.SE - Show the structures of the fragments you would...Ch. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - Write molecular formulas for compounds that show...Ch. 12.SE - Camphor, a saturated monoketone from the Asian...Ch. 12.SE - The nitrogen rule of mass spectrometry says that a...Ch. 12.SE - In light of the nitrogen rule mentioned in Problem...Ch. 12.SE - Nicotine is a diamino compound isolated from dried...Ch. 12.SE - The hormone cortisone contains C, H, and O, and...Ch. 12.SE - Halogenated compounds are particularly easy to...Ch. 12.SE - Prob. 22APCh. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - 2-Methylpentane (C6H14) has the mass spectrum...Ch. 12.SE - Assume that you are in a laboratory carrying out...Ch. 12.SE - What fragments might you expect in the mass...Ch. 12.SE - How might you use IR spectroscopy to distinguish...Ch. 12.SE - Would you expect two enantiomers such as...Ch. 12.SE - Would you expect two diastereomers such as meso-2,...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - How could you use infrared spectroscopy to...Ch. 12.SE - Prob. 32APCh. 12.SE - At what approximate positions might the following...Ch. 12.SE - How would you use infrared spectroscopy to...Ch. 12.SE - At what approximate positions might the following...Ch. 12.SE - Assume that you are carrying out the dehydration...Ch. 12.SE - Assume that you are carrying out the base-induced...Ch. 12.SE - Prob. 38APCh. 12.SE - Carvone is an unsaturated ketone responsible for...Ch. 12.SE - Prob. 40APCh. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - 4-Methyl-2-pentanone and 3-methylpentanal are...Ch. 12.SE - Grignard reagents undergo a general and very...Ch. 12.SE - Ketones undergo a reduction when treated with...Ch. 12.SE - Nitriles, R–=C≡N, undergo a hydrolysis...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - Prob. 50AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- If you know the energies of the HOMO and LUMO of a particular compound, how could you calculate the lowest energy optic absorption?arrow_forwardCalculate the IHD of C7H6XNO and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.arrow_forwardWhat is the structure from the formula C10H12O and the spectra?arrow_forward
- Morphine, heroin, and oxycodone are three addicting analgesic narcotics. How could IR spectroscopy be used to distinguish these three compounds from each other?arrow_forwardIdentify the important absorption peaks in the following IR spectra and describe the appearance of the important absorption peaks in the IR Spectra. No cursive writing please. Thank youarrow_forwardIdentify the important absorption peak and its description in the IR spectra.arrow_forward
- 22. Which of the following compounds gives an infrared Spectrum with peaks at 3300cm (sharp Peak) and 2150 cm (sharp Peak) ? H LH ₂ CH ₂ C = CH CH3C=CCH 3 2 1 HCCH H₂C A) I B)2 c) 3 04 CAZ H 9 CH₂arrow_forwardHow does the operating frequency in NMR spectroscopy compare with the operating frequency in IR and UV/Vis spectroscopy?arrow_forwardWhich molecule has the IR spectrum below. A B C D A Transmitance 1 0.8 0.6 0.4 0.2 3000 B INFRARED SPECTRUM 2000 Wavenumber (cm-1) C 1000 D 'Нarrow_forward
- A certain compound has the following molecular formula C4H6 and ha sthe ollowing IR spectra. Use the file response to upload the structure. Explain 4000 3000 m 1500 Wavenumbers (cm-¹) 2000 1000 500arrow_forwardWhat is molecular spectroscopy and UV-VIS absorption, IR absorption spectroscopy and fluorescence spectroscopy?arrow_forwardChoose the compound with the broadest IR signal between 3600 cm1 and 3000 cm1 OH HO OH HO A D B.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY