
Interpretation:
The condensed formulas for three
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Condensed structure:
Condensed structure is the line diagram, the carbon-carbon bonds are represented with lines, and hydrogens are absent entirely.
Carbonyl group:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with two alkyl or aryl group is called as ketone.
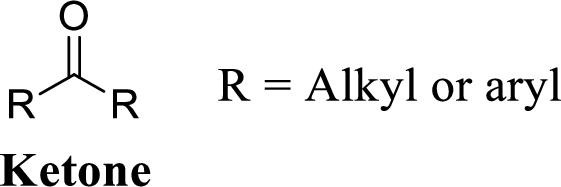
Trending nowThis is a popular solution!

Chapter 1 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Draw condensed structural formulas for all compounds with the molecular formula C4H8O that contain a carbonyl group (there are two aldehydes and one ketone).arrow_forwardStructural formula for the four ketones with the molecular formula C6H12O.arrow_forwardDraw condensed structural formulas for the two aldehydes with the molecular formula C4H8Oarrow_forward
- Draw condensed structural formulas for all compounds with the molecular formula C4H8O that contain Q.) A carbonyl group (there are two aldehydes and one ketone).arrow_forwardDraw Structural formulas to the eight carboxylic acids with the molecular formula C6H12O2.arrow_forwardDraw Lewis structures and condensed structural formulas for the four alcohols with the molecular formula C4H10O. Classify each alcohol as primary, secondary, or tertiary.arrow_forward
- An organic compounds having molecular formula C5H2O, which of the following is correct for this compound: O can be aldehyde and ketone O can be alcohol and aldehyde O can be alcohol and ether O can be ether and ketonearrow_forwardDescribe the difference between an aldehyde and a ketone, and indicate how each differs from an alcohol.arrow_forwardStructural formula for the eight carboxylic acids with the molecular formula C6H12O2.arrow_forward
- What is the enthalpy change to convert one mole of methane to methanol?arrow_forwardWe see that 1-propanol and 2-propanol have the same molecular formula, C3H7OH, but different molecular structures. What is the name for molecules that have the same molecular formula but different structural formulas (different shapes)? Use the specific term.arrow_forwardStructural formula for the four alcohols with the molecular formula C5H12O.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

