
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
4th Edition
ISBN: 9781119234722
Author: Klein
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1.3, Problem 1.10P
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
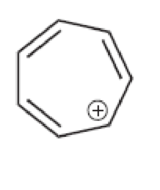
Answer: _____
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10. Identify the following as aromatic, non-aromatic, or anti-aromatic. List answers below.
N'
Classify each of the following compounds as aromatic or non-aromatic.
SH
D
C
Structure A
V[Choose ]
Aromatic
Non-aromatic
Structure B
Choose]
Structure C
[Choose ]
Structure D
[ Choose )
Structure E
[ Choose]
Structure F
Choose1
B.
I need help identifying which compounds are aromatic, antiaromatic, non aromatic. Please help
Chapter 1 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
Ch. 1.2 - Prob. 1.2PCh. 1.2 - Prob. 1.3PCh. 1.2 - Prob. 1.4PCh. 1.2 - Prob. 1.5PCh. 1.2 - Prob. 1.6PCh. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
61. What is the pH of a solution in which 224 mL of HCl(g), measured at 27.2 °C and 1.02 atm, is dissolved in 1...
Chemistry: A Molecular Approach (4th Edition)
Predict whether a precipitation reaction will occur when aqueous solutions of the following substances are mixe...
CHEMISTRY-TEXT
Two things to be done to change the concentration of the products of the following reaction. 2NO2 (g) ⇌ N2...
Living By Chemistry: First Edition Textbook
a. Find potassium (K) in the periodic table and predict how many valence electrons it has. b. What orbital does...
Essential Organic Chemistry (3rd Edition)
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ô https://app.101edu.co Question 3 of 24 A substance which is cyclic, conjugated, and has 4n + 2 pi electrons but contains a sp³ hybridized atom in the ring is classified as A) aromatic. B) non-aromatic. C) anti-aromatic. e here to search 82% 54°F C F5 F8 F9 F10 F11 F12 F3 F4 F6 F7 %23 $4 & 3. 4. 6. 17 8. 9. Y U H JK Larrow_forwardAromaticity and Properties Identify the properties that describe aromatic compounds. Aromatic Answer Bank planar all single bonds flexible rigid twisted alternating double and single bonds electrons fully delocalized in a ring all bond lengths between double and single bond lengths all double bonds reactive unpaired electrons very unstable very stablearrow_forwardDetermine if the following molecules are aromatic, non-aromatic, or anti-aromatic.arrow_forward
- Determine if the following compounds are not aromatic, aromatic or anti-aromatic. :O: 00 IZ:arrow_forwardDetermine whether the following compounds are Aromatic, Antiaromatic, or Nonaromatic.arrow_forwardClassify each of the following molecules as aromatic, antiaromatic, or nonaromatic. O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromaticarrow_forward
- Periodic Table Calculator 7 of 24 Chemistry: Fundamentals and Principles preserted by Saplng Learning Vanillin, CeHeO3, is the active ingredient in vanilla flavoring. It contains a six-membered aromatic ring with an aldehyde group on carbon 1, an alkoxy group (ether) on carbon 3, and a hydroxyl group (alcohol) on carbon 4. Draw the structure of vanillin. e Previous Check Answer Next Hint about us careers privacy policy terms 2019 End of Seaso.doc lyparrow_forwardplease identify if Aromatic, anti-aromatic, or non-aromatic?arrow_forwardPart A CH,- Spell out the full name of the alkyl group. Submit Request Answer Part B CH3CH2CH(CH3)- Spell out the full name of thg alkyl group. Submit Request Answer Provide Feedbackarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY