
Concept explainers
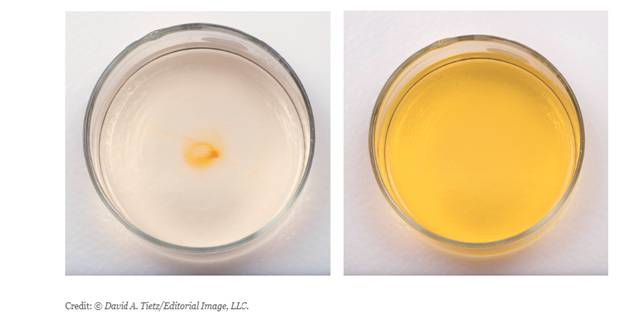
Iodine
Describe the change in solubility of I2 in terms of the change in intermolecular forces.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Chemistry
- In a solution with carbon tetrachloride as the solvent, the compound VCl4. undergoes dimerization: 2VCl4V2Cl8 When 6.6834 g VCl4. is dissolved in 100.0 g carbon tetrachloride, the freezing point is lowered by 5.97C. Calculate the value of the equilibrium constant for the dimerization of VCl4 at this temperature. (The density of the equilibrium mixture is 1.696 g/cm3, and Kf = 29.8C kg/mol for CCl4.)arrow_forwardThe following data table indicates the solubility of a substance. By examining the data presented, predict the solubility of the substance at 50 oC. Temperature (oC) Solubility (mM) 5 5.00 15 4.35 25 3.70 35 3.05 45 2.40 2.00 mM 1.40 mM 1.00 mM 2.10 mM 1.75 mMarrow_forwardThe solubility of Fe(OH)2 is 2.1 x 10-4 g L-1. What is the Ksp for Fe(OH)2 at 25 °C?arrow_forward
- Strontium fluoride is used to make optical material to produce devices due to its high conductivity and its optical properties similar to calcium fluoride. Strontium fluoride has the ability to transmit ultraviolet and infrared waves, which is why it is used in glass, windows, and lenses. Irritating to eyes and skin, and is toxic if inhaled or swallowed. If the Kps of strontium fluoride at 25 ° C is 2.90 × 10−9: a) Determine the molar solubility at 25 ° C. b) What are the fluoride ion concentration, in mol / L, in a saturated strontium fluoride solution? c) If two solutions are mixed until reaching 1.00 L with a fluoride concentration of 2.34x10-4mol / L and a strontium concentration of 7.81x10-3, explain whether or not a precipitate will form.arrow_forwardThe solubility of silver phosphate, Ag3PO4 at 25°C is 1.61 x 10-5 mol/L. Determine the concentration of the Ag+ ion in a saturated solution.arrow_forwardAt 25 °C the solubility of NaCl is 36.0g/100g H2O How much NaCl can dissolve in 450 g of water at that temperature?arrow_forward
- 7.A solution is prepared by dissolving 0.030 mol of potassium perchlorate in 75 mL of hot water. KClO4 dissolves according to the equation KClO4 (s) ? K+ (aq) + ClO4- (aq). Will a precipitate form if the solution is cooled to 25oC? Assume that a negligible change in volume occurs during the operations. (Ksp = 8.9 x 10-3).arrow_forwardThe Ksp of Barium Carbonate, BaCO3, is 2.58x10^-9. Calculate the solubility of this compound in g/L.arrow_forwardThe solubility of TlCl [thallium(I) chloride], an intermediate formed when thallium is being isolated from ores, is 3.12 grams per liter at 20 °C. What is its solubility product?arrow_forward
- What is the solubility of La(IO3)3 in a solution that contains 0.100 M IO3- ions? (Ksp of La(IO3)3 is 7.5 x 10^-12?arrow_forwardAt 25.0∘C, the molar solubility of calcium phosphate in water is 1.10×10−7 M. What is the solubility of calcium phosphate in parts per million? Express your answer in parts per million to three significant figures.arrow_forwardThe solubility of aqueous PbCr2O7 at 25 oC is 1.55 x 10–2 g/L at 25.0 oC. Calculate the Ksp of PbCr2O7 at this temperature. MM PbCr2O7 = 423.188 g/molarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





