
Concept explainers
Interpretation:
The missing reagents for each step in Your Turn 13.6 are to be supplied.
Concept introduction:
In order to identify the missing reagents in the given reaction sequence, it is important to identify if the reaction involves a functional group transformation, or it is a reaction that alters the carbon skeleton. In each given reaction, the bonds which are broken and formed are to be identified. The regioselectivity and stereochemistry play an important role in the outcome of the reaction when a specific reagent is used.
Under basic conditions, the nucleophile attacks the
Answer to Problem 13.1P
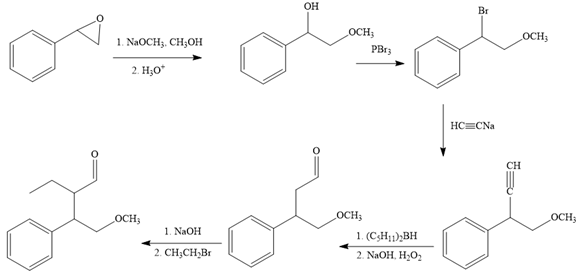
The missing reagents for each step in the given reaction sequence are given below:
Explanation of Solution
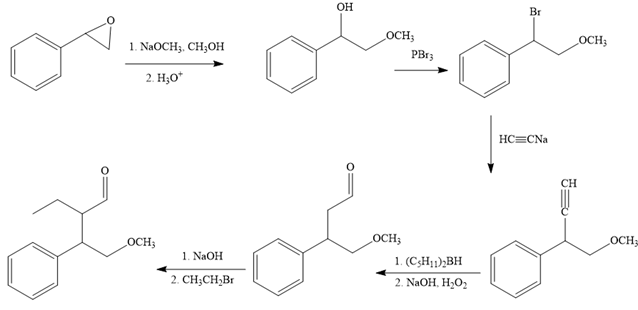
The reaction sequence given in Your Turn 13.6 is:
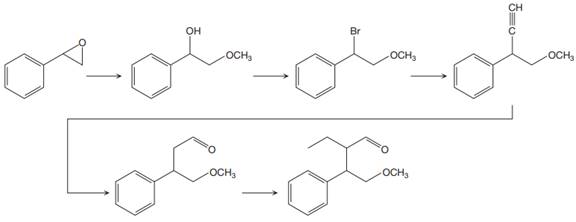
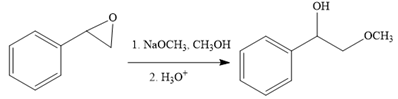
The first reaction is the conversion of an epoxide to alcohol. Thus, it is a functional group transformation reaction in which the nucleophile,
The first reaction and the missing reagents for it are shown below:
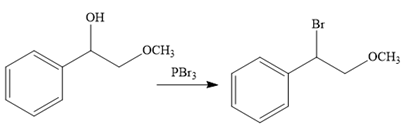
The second reaction also involves functional group transformation. The alcoholic (
In the third reaction, the bromine atom is replaced by an acetylene group (
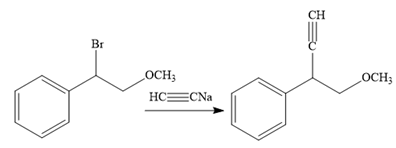
The fourth reaction in the given reaction sequence is a reaction involving a functional group transformation. Terminal alkynes undergo a hydroboration-oxidation reaction which leads to the formation of an aldehyde. The reagents used in the hydroboration-oxidation reaction are disiamylborane [
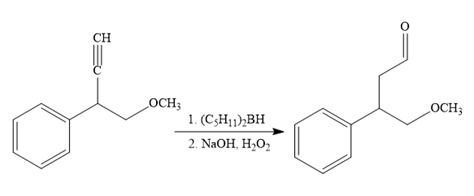
The fifth reaction involves the alteration of the carbon skeleton. The alpha hydrogen attached to an alpha carbon in aldehydes is weakly acidic. A strong base abstracts this alpha hydrogen to form an enolate ion. This enolate serves as a nucleophile and reacts with an alkyl halide via
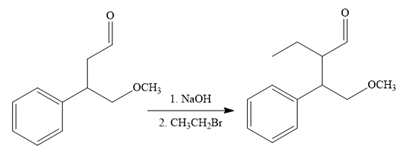
The complete reaction sequence with appropriate reagents for each are given below:
In order to identify the missing reagents in the given reaction sequence, it is important to identify if the reaction involves a functional group transformation or it is a reaction that alters the carbon skeleton.
Want to see more full solutions like this?
Chapter 13 Solutions
ORGANIC CHEMISTRY PRINCIPLES & MECHANISM
- 1. Draw the starting structure that would lead to this major product 2. Draw the major product of this reaction. Ignore inorganic 3. Draw the major product of this reaction. Ignore inorganic under these conditions. byproducts. byproducts. H3O* H3O* H3O* НОarrow_forwardI am very confused with this please help me .arrow_forwardCircle the correct bolded word. a. Hydrogenation of an alkyne with palladium on carbon can / cannot be controlled to give an alkene. b. When a reaction becomes more exothermic, the transition state looks more like the substrate / product. C. Addition of two equivalents of HBr to an alkyne results in a vicinal / geminal dibromide. d. An SN2 reaction involves the collision of two alkyl halides to form a new carbon-carbon bond. True / Falsearrow_forward
- 4. Provide a synthetic route to the following molecule using benzene and cyclohexane. Reagents cannot contain more than one carbon. Provide a mechanism for the last step of your synthetic route.arrow_forwardHO Cro3 H2S04 Edit Click on the drawing box above to activate the MarvinSketch drawing tool and then draw your answer to this question. If there is no reaction, then check the "no reaction" box below. no reactionarrow_forward6. Multi Step Synthesis. Propose a synthetic route from the reactant to the product using any reagents you need. Nalt LINH4arrow_forward
- Create a Synthetic Route Fill in the missing information that describes how to synthesize the target molecule from the starting molecule. Be aware that this is a multi-step synthesis, and that you have not been told how many steps are required. Work backward from the target molecule one step at a time. To complete a step, you must correctly identify both the reaction conditions and the starting molecule for that particular step. Target molecule: Starting molecule: aldehyde butyraldehyde alkane Show reaction map for: Chapters 6-10arrow_forwardSynthesis. Design a synthetic route to perform the transformations shown below. You may use any other reagents necessary, as long as the starting material is incorporated into the product.arrow_forward2. Draw the structures and explain why CH3CH₂O and CH3CO₂ are good nucleophiles but CH3SO3, water, and alcohols (R-OH) are poor nucleophiles. Propose a 'cutoff' for the amount of negative charge needed to be a good nucleophile. CH3CH₂O CH3CO₂ CH3SO3 H₂O CH₂OHarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
