
Interpretation:
Nitrogen containing functional group has to be labelled in lidocaine and also they have to be classified as primary, secondary or tertiary.
Concept Introduction:
Amides contain a nitrogen atom that is bonded to carbonyl group. The nitrogen atom that is bonded to the carbonyl group may be bonded to alkyl groups or hydrogen atoms. Depending on the number of carbon atoms that is attached to the nitrogen atom, the amide is classified as primary, secondary, and tertiary.
Primary amide is the one that contains only one
Explanation of Solution
Structure of lidocaine is:
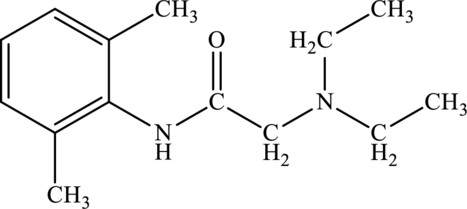
Lidocaine contains two nitrogen atoms. Among this, one nitrogen atom is bonded to a carbonyl group carbon atom and another nitrogen atom is bonded to carbon atoms. Thus the compound contains amine and amide functional group. This is labelled as follows.
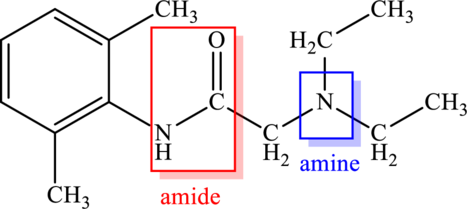
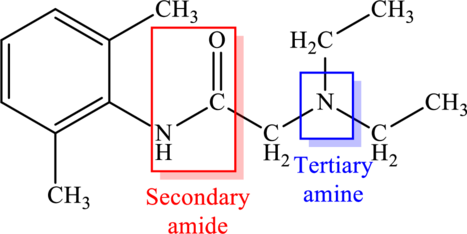
Nitrogen atom that is present in amide is bonded to two carbon atoms. This means there are two
Want to see more full solutions like this?
Chapter 13 Solutions
PRIN.OF GENERAL,ORGANIC+BIOLOG.CHEM.
- Draw a structural formula for each amine and amine derivative. Q.) trans-2-Aminocyclohexanolarrow_forwardWhat ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forwardIdentify each functional group located in the following rings. Whichstructure represents a lactone—a cyclic ester—and which represents a lactam—a cyclic amide?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,



