
Which of the following species will not have permanent dipole moments? (a) Hydrogen cyanide,
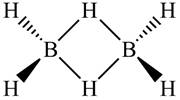
Where the bridging hydrogens are perpendicular to the four terminal hydrogens, which are all coplanar
Trending nowThis is a popular solution!

Chapter 13 Solutions
EBK PHYSICAL CHEMISTRY
- 4. (a) Draw the shape of the atomic valence orbitals formed by the overlaping of two fluoride 2p atomic orbitals. (b) Draw the molecular orbital diagrams for F2 and F2*. Identify their bond order and magnetic properties. (c) An unstable nucleus exhibit radioactivity. (i) Explain how the number of protons and neutrons in a radioactive nucleus can be used to predict its probable mode decay. (ii) Illustrate your answer in (i) with a schematic graph.arrow_forwardFrom their Lewis structures, determine the number of sand π bonds in each of the following molecules or ions:(a) CO2; (b) cyanogen,(CN2); (c) formaldehyde, H2CO;(d) formic acid, HCOOH, which has one H and two O atomsattached to C.arrow_forwardBoric acid, B(OH)3 (or H3BO3), does not lose a proton in water, but rather bonds to the O atom of an H2O molecule, which then releases an H+ ion to form the B(OH)4- ion. Use VESPR theory to draw structures, with ideal bond angles, for boric acid and the anion it forms when it reacts with water.arrow_forward
- The standard potential of a redox couple is a measure of the thermodynamic tendency of an atom, ion, or molecule to accept an electron (Topic 6D). Studies indicate that there is a correlation between the LUMO energy and the standard potential of aromatic hydrocarbons. Do you expect the standard potential to increase or decrease as the LUMO energy decreases? Explain your answer.arrow_forwardThe dipole moment of gaseous Ag – Cl is ? = 6.08 D, and the bond distance is r(Ag − Cl) = 228. 1 pm. Calculate the percent ionic character for this bond in the gas phase. In solid form it is however, found the following values for the two silver halides Ag − CL and Ag − F: See image Explain why solid silver chloride is sparingly soluble in water, while solid silver fluoride is moderately soluble B) Explain why solid silver chloride is sparingly soluble in water, while solid silver fluoride is moderately soluble Given extra information: Formal charge: ?? = ??? – (??? + ???); where ??? = valence electrons in free atom, ???= free-pair electrons, ??? = number of bonds to the atom. The elementary charge: ? = 1.6022 × 10−19 C.1 Debye (?)= 3.3356 × 10−30 C × m. The dipole moment has definition: ? = ? × ?, where ? is partialcharge in Coulomb (?), ? is the bond distance (?). Picometer: 1 ?? = 1 × 10−12arrow_forwardThe HI molecule has an equilibrium bond length of 1.59 Angstroms and a dipole moment of 0.42 Debye. IF one were to treat this molecule as a pair of point charges +-q separated by the same distance as its bond length, what would the magnitude of q need to be to give the experimental dipole moment? Express your answer as q divided by the proton charge. In other words, what fraction of a full proton charge is the charge magnitude of the ion pair?arrow_forward
- Thionyl chloride (SOCl₂) is a sulfur oxohalide used indus-trially to dehydrate metal halide hydrates. (a) Write a balancedequation for its reaction with magnesium chloride hexahydrate,in which SO₂ and HCl form along with the metal halide. (b) Drawa Lewis structure of SOCl₂ with minimal formal charges.arrow_forward(a) Find the angle u between adjacent nearest-neighbor bonds in the silicon lattice. Recall that each silicon atom is bonded to four of its nearest neighbors.The four neighbors form a regular tetrahedron— a pyramid whose sides and base are equilateral triangles. (b) Find the bond length, given that the atoms at the corners of the tetrahedron are 388 pm apart.arrow_forwardThe sulfate ion can be represented with four S-O bonds or with two S-O and two So=O bonds.(a) Which representation is better from the standpoint of formal charges?(b) What is the shape of the sulfate ion, and what hybrid orbitals of S are postulated for the σ bonding?(c) In view of the answer to part (b), what orbitals of S must be used for the π bonds? What orbitals of O?(d) Draw a diagram to show how one atomic orbital from S and one from O overlap to form a π bond.arrow_forward
- Propylene, C3H6, is a gas that is used to form the importantpolymer called polypropylene. Its Lewis structure is given. (a) What is the total number of valence electrons in the propylenemolecule? (b) How many valence electrons are usedto make σ bonds in the molecule? (c) How many valenceelectrons are used to make π bonds in the molecule? (d) Howmany valence electrons remain in nonbonding pairs in themolecule? (e) What is the hybridization at each carbon atomin the molecule?arrow_forward2(a) Provide the Lewis structures for both CH3OH and C2H3Cl. 2(b) What is the largest bond angle among all the bond angles in CH3OH and C2H3Cl? Listthe three atoms making this largest bond angle, and estimate the value of the angle.2(c) What intermolecular forces are present(i) between CH3OH molecules?(ii) between C2H3Cl molecules?arrow_forward(a) What are trihalomethanes (THMs)? (b) Draw the Lewisstructures of two example THMs.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
