
Package: Organic Chemistry With Connect 2-semester Access Card
4th Edition
ISBN: 9781259671838
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.39P
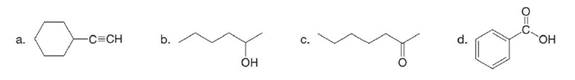
What major IR absorptions are present above
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What major IR absorptions are present above 1500 cm−1 for each compound?
What types of bonds are responsible for the absorptions above 1500 cm−1 in the IR spectra for compounds A and B?
What major IR absorptions are present above 1500 cm−1 for attached compound ?
Chapter 13 Solutions
Package: Organic Chemistry With Connect 2-semester Access Card
Ch. 13 - What is the mass of the molecular ion formed from...Ch. 13 - Prob. 13.2PCh. 13 - Prob. 13.3PCh. 13 - What molecular ions would you expect for the...Ch. 13 - The mass spectrum of 2,3-dimethylpentane also...Ch. 13 - The base peak in the mass spectrum of 2, 2,...Ch. 13 - (a) What mass spectral fragments are formed by ...Ch. 13 - What cations are formed in the mass spectrometer...Ch. 13 - The low-resolution mass spectrum of an unknown...Ch. 13 - Benzene, toluene, and p-xylene BTX are often added...
Ch. 13 - Prob. 13.11PCh. 13 - Prob. 13.12PCh. 13 - Prob. 13.13PCh. 13 - Prob. 13.14PCh. 13 - How do the IR spectra of the isomers cyclopentane...Ch. 13 - How do the three isomers of molecular formula...Ch. 13 - Problem 13.18 What functional groups are...Ch. 13 - Problem-13.19 What are the major IR absorptions in...Ch. 13 - Problem-13.20 What are the major IR absorptions in...Ch. 13 - Prob. 13.20PCh. 13 - Problem-13.22 Propose structures consistent with...Ch. 13 - 13.23 What major IR absorptions are present above ...Ch. 13 - Problem-13.24 The mass spectrum of the following...Ch. 13 - What molecular ion is expected for each compound?Ch. 13 - Which compound gives a molecular ion at m/z= 122,...Ch. 13 - Propose two molecular formulas for each molecular...Ch. 13 - Propose four possible structures for a hydrocarbon...Ch. 13 - Match each structure to its mass spectrum. a. b....Ch. 13 - 13.32 Propose two possible structures for a...Ch. 13 - 13.33 What cations are formed in the mass...Ch. 13 - 13.35 For each compound, assign likely...Ch. 13 - Prob. 13.32PCh. 13 - 13.37 Propose a structure consistent with each...Ch. 13 - 13.38 A low-resolution mass spectrum of the...Ch. 13 - Can the exact mass obtained in a high-resolution...Ch. 13 - 13.39 Primary alcohols often show a peak in their...Ch. 13 - 13.40 Like alcohols, ethers undergo α cleavage by...Ch. 13 - Which of the highlighted bonds absorbs at higher v...Ch. 13 - What major IR absorptions are present above...Ch. 13 - How would each of the following pairs of compounds...Ch. 13 - 13.44 Morphine, heroin, and oxycodone are three...Ch. 13 - Prob. 13.42PCh. 13 - 13.47 Match each compound to its IR spectrum
Ch. 13 - 13.48 Propose possible structures consistent with...Ch. 13 - A chiral hydrocarbon X exhibits a molecular ion at...Ch. 13 - 13.50 A chiral compound has a strong absorption...Ch. 13 - 13.51 Treatment of benzoic acid with followed by...Ch. 13 - 13.52 Treatment of benzaldehyde with in aqueous ...Ch. 13 - Prob. 13.49PCh. 13 - 13.54 Reaction of 2-methylpropanoic acid with ...Ch. 13 - 13.55 Reaction of pentanoyl chloride with lithium...Ch. 13 - Prob. 13.52PCh. 13 - 13.57 Treatment of anisole with and forms P,...Ch. 13 - 13.58 Reaction of with forms compound ,...Ch. 13 - Problem-13.59 The carbonyl absorption of an amide...Ch. 13 - Prob. 13.56PCh. 13 - Problem-13.61 Explain why a ketone carbonyl...Ch. 13 - 13.62 Oxidation of citronellol, a constituent of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- This is the 13C spectrum for an X compound with molecular formula C7H12O4. The substance is not soluble in NaHCO3 and has a stretch at 1740cm-1 on the IR spectrum. What is the structure of X?arrow_forwardwhat does it mean if a compound has a peak that has medium stretch at 1658cm-1 in an IR spectra? Does it mean there is a possiblity that the compound could be a ketone?arrow_forwardWhat is the difference between absorption frequencies (infrared spectroscopy) for 2,3-dimethylbut-1-ene and 2,3-dimethylbut-2-ene? Why is it that the first structure has moderate intensity for the C=C bond while the second structure has weak/non-existent intensity for the "same" C=C bond?arrow_forward
- Propose a structure consistent with each set of data. C8H10 : IR absorptions at 3108–2875, 1606, and 1496 cm−1arrow_forwarda) what bond is present in ketones but not alkanes? b) what is the wavenumber range of the signals that are present in the diagnostic region of all of the ketone IR spectra (but not the alkanes)? c) is the new band strong or weak compared with a C=C or a C≡C stretch?arrow_forwardA compound has a molecular ion in its mass spectrum at m / z = 58, and a strong absorption band at its IR at 1720. What would be the structure?arrow_forward
- Carbonyl (C=O) group of acetone stretches at 1724 cm-1 in IR but the same group stretches at much lower (1691 cm-1) wave number for 3-Nitroacetophenone. Why?arrow_forwardHow could 1H NMR spectroscopy be used to distinguish betweencompounds X and Y?arrow_forwardThe 1H NMR spectrum of 1,2-dimethoxyethane (CH3OCH2CH2OCH3) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downeld from TMS. (a) Calculate the chemical shift of each absorption. (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY