
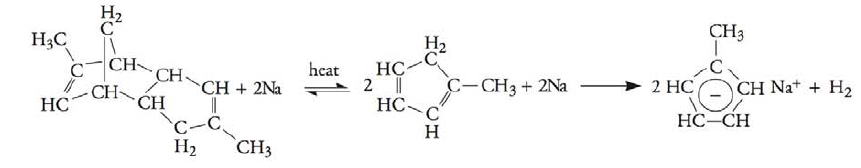
On December 19, 2007, the T2 Laboratories, Inc., reactor exploded in a runaway reaction. The reaction of methyl cyclopentadienyl dimer and sodium produces sodium methyl cyclopentadiene and hydrogen:
The reactor has to be cooled when its temperature reaches
If the rare constant for a typical reaction is
if all the heat were released at once?
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK STUDY GUIDE TO ACCOMPANY CHEMISTRY:
Additional Science Textbook Solutions
Basic Chemistry (5th Edition)
Chemistry: The Central Science (14th Edition)
Inorganic Chemistry
Living by Chemistry
Chemistry For Changing Times (14th Edition)
Essential Organic Chemistry (3rd Edition)
- Silicon forms a series of compounds analogous to the al-kanes and having the general formula SinH2n+2. The first of these compounds is silane, SiH4, which is used in the electronics industry to produce thin ultrapure silicon films. SiH4(g) is somewhat difficult to work with because it is py-ropboric at room temperature—meaning that it bursts into flame spontaneously when exposed to air. (a) Write an equation for the combustion of SiH4(g). (The reaction is analogous to hydrocarbon combustion, and SiO2 is a solid under standard conditions. Assume the water produced will be a gas.) (b) Use the data from Appendix E to calculate ? for this reaction. (c) Calculate G and show that the reaction is spontaneous at 25°C. (d) Compare G for this reaction to the combustion of methane. (See the previous problem.) Are the reactions in these two exercises enthalpy or entropy driven? Explain.arrow_forwardAmoxicillin is an antibiotic packaged as a powder. When it is used to treat babies and small animals, the pharmacist or veterinarian must suspend it in water, so that it can be administered orally with a medicine dropper. The label says to dispose of unused suspension after 14 days. It also points out that refrigeration is required. In the context of this chapter, what is implied in the latter two statements?arrow_forwardSubstances that poison a catalyst pose a major concern for many engineering designs, including those for catalytic converters. One design option is to add materials that react with potential poisons before they reach the catalyst. Among the commonly encountered catalyst poisons are silicon and phosphorus, which typically form phosphate or silicate ions in the oxidizing environment of an engine. Group 2 elements are added to the catalyst to react with these contaminants before they reach the working portion of the catalytic converter. If estimates show that a catalytic converter will be exposed to 625 g of silicon during its lifetime, what mass of beryllium would need to be included in the design?arrow_forward
- Can a reaction mechanism ever be proven correct? Can it be proven incorrect?arrow_forwardPeroxynitric acid (HOONO2) is an unstable molecule that decomposes to nitric acid and oxygen: 2HOONO2(aq) → 2HNO3(aq) + O2(g)When the concentration of peroxynitic acid is graphed against time, the resulting plot is curved, but if the logarithm of this concentration is plotted, we instead get a straight line. Based on this, which statement is true? a) This decay is a second order in peroxynitric acid. b) The slope of the straight-line graph is the rate constant. c) One needs the concentration of peroxynitric acid to calculate its half-life. d) The rate law appears to be of the form -Δ[HOONO2]/Δt = k[HOONO2].arrow_forwardConsider the following reaction mechanism: Step 1: A3(g) → A2(g) + A(g) Step 2: A3(g) + A(g) → 2A2(g) What is the role of A(g) and what is the overall balanced equation? Periodic Table and Datasheet O A(g) is an intermediate; 2A3(g) + A(g) → 3A2(g) + A(g) O A(g) is a catalyst; 2A3(g) + A(g)→ 3A2(g) + A(g) O A(g) is an intermediate; 2A3(g) –→3A2(g) O A(g) is a catalyst; 2A3(g) → 3A2(g)arrow_forward
- Scientists can determine the age of ancient objects by the method of radiocarbon dating. The bombardment of the upper atmosphere by cosmic rays converts nitrogen to a radioactive isotope of carbon, 14C, with a half-life of about 5,730 years. Vegetation absorbs carbon dioxide through the atmosphere, and animal life assimilates 14C through food chains. When a plant or animal dies, it stops replacing its carbon, and the amount of 14C present begins to decrease through radioactive decay. Therefore, the level of radioactivity must also decay exponentially. Dinosaur fossils are often dated by using an element other than carbon, like potassium-40, that has a longer half-life (in this case, approximately 1.25 billion years). Suppose the minimum detectable mass is 0.4% and a dinosaur is dated with 40K to be 67 million years old. Is such a dating possible? What is the maximum age (in millions of years) of a fossil that could be dated using 40K? (Round your answer to one decimal place.) (Can you…arrow_forwardScientists can determine the age of ancient objects by the method of radiocarbon dating. The bombardment of the upper atmosphere by cosmic rays converts nitrogen to a radioactive isotope of carbon, 14C, with a half-life of about 5,730 years. Vegetation absorbs carbon dioxide through the atmosphere, and animal life assimilates 14C through food chains. When a plant or animal dies, it stops replacing its carbon, and the amount of 14C present begins to decrease through radioactive decay. Therefore, the level of radioactivity must also decay exponentially. a) Dinosaur fossils are too old to be reliably dated using carbon-14. Suppose we had a 67 million year old dinosaur fossil. What percent of the living dinosaur's 14C would be remaining today? (Round your answer to five decimal places.) b) Suppose the minimum detectable amount is 0.7%. What is the maximum age (in years) of a fossil that we could date using 14C? (Round your answer to the nearest year.)arrow_forwardScientists can determine the age of ancient objects by the method of radiocarbon dating. The bombardment of the upper atmosphere by cosmic rays converts nitrogen to a radioactive isotope of carbon, 14C, with a half-life of about 5,730 years. Vegetation absorbs carbon dioxide through the atmosphere, and animal life assimilates 14C through food chains. When a plant or animal dies, it stops replacing its carbon, and the amount of 14C present begins to decrease through radioactive decay. Therefore, the level of radioactivity must also decay exponentially. Dinosaur fossils are too old to be reliably dated using carbon-14. Suppose we had a 66 million year old dinosaur fossil. What percent of the living dinosaur's 14C would be remaining today? (Round your answer to five decimal places.) 0.0000007537 X % Suppose the minimum detectable amount is 0.2%. What is the maximum age (in years) of a fossil that we could date using 14C? (Round your answer to the nearest year.) yrarrow_forward
- 89. At 700 K acetaldehyde decomposes in the gas phase to methane and carbon monoxide. The reaction is: CH3CHO(g) CH4(g) + CO(g) A sample of CH3CHO is heated to 700 K and the pressure is measured as 220 mbar before any reaction takes place. The kinetics of the reaction are then followed by measurements of total pressure and these data are obtained: t (s) PTotal (mbar) 0 220 1000 240 3000 270 7000 310 4 Find the rate law, the specific rate constant, and the total pres- sure after 2.00 × 104 s.arrow_forward09:00 Edit סם ⠀ 2 Assignment 3 (NCHE312) The total pressure of the following system is used to monitor the progress of the chemical reaction. SO₂Cl2(g) → SO₂(g) + Cl₂(g) The reaction is initiated, and the following data are obtained: 0 3 6 9 12 15 Time (h) PTotal (kPa) 14.79 17.26 18.90 11.07 19.99 20.71 Pso₂Cl₂ Pso₂ Pclz Additional information: As the reaction proceeds, stoichiometry dictates that for every mole of SO₂Cl₂ that dissociates one mole of each of SO₂ and Cl₂ is produced. Defining the extent of dissociation/reaction as E, and the initial pressure as Po, the total pressure is given by: Ptotal = Pso₂Cl₂ + Ps0₂ + Pcl₂ = (Po-5 + 5 + 5 a) Calculate the pressure of each component at each time interval, i.e. complete the above table. b) Use a graphical method and determine what the order of the reaction is with respect to SO₂Cl₂? (i) Show the plot as well as the linear fit of the data. (ii) Motivate your answer. c) Determine the rate constant for this reaction. d) Give the rate…arrow_forwardOne of the least abundant nitrogen oxides in the atmosphere is dinitrogen pentoxide. One reason concentrations of this oxide are low is that the molecule is unstable and rapidly decomposes to N2O4 and O2: 2N2O5(g) → 2N2O4(g) + O2(g) A kinetic study of the decomposition of N2O5 at a particular temperature yielded the data in the following Table. Assume that the decomposition of N2O5 is first order in N2O5. Test the validity of your assumption. Determine the value of the rate constant.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




