
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781337598255
Author: Spencer L. Seager
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.42E
Assign the IUPAC name to each of the following ethers.
Name the smaller alkyl group as the alkoxy group.
a.
b. 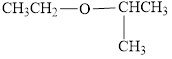
c. 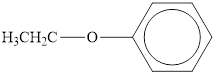 d.
d. 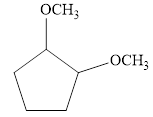
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3l
Identify the IUPAC name of the given structure.
A. 2 - propylpropanal
B. hexanal
C. 2 - methylpentanal
1. What does the ff compound belong to:
a. ketone
b. alcohol
c. aldehyde
d. alkane
2. What group does the ff organic compound belong?
a. ketone
b. carboxylic acid
c. aldehyde
d. alkyl halide
3. What group does the ff organic compound belong?
a. alkene
b. ketone
c. alcohol
d. aldehyde
Chapter 13 Solutions
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
Ch. 13 - Draw general formulas for an alcohol and phenol,...Ch. 13 - Prob. 13.2ECh. 13 - Assign IUPAC names to the following alcohols: a....Ch. 13 - Assign IUPAC names to the following alcohols: a....Ch. 13 - Several important alcohols are well known by...Ch. 13 - Prob. 13.6ECh. 13 - Draw structural formulas for each of the...Ch. 13 - Draw structural formulas for each of the...Ch. 13 - Name each of the following as a derivative of...Ch. 13 - Name each of the following as a derivative of...
Ch. 13 - Prob. 13.11ECh. 13 - Draw structural formulas for each of the...Ch. 13 - Prob. 13.13ECh. 13 - Classify the following alcohols as primary,...Ch. 13 - Classify the following alcohols as primary,...Ch. 13 - Draw structural formulas for the four aliphatic...Ch. 13 - Why are the boiling points of alcohols much higher...Ch. 13 - Arrange the compounds of each group in order of...Ch. 13 - Prob. 13.19ECh. 13 - Draw structural formulas for the following...Ch. 13 - Prob. 13.21ECh. 13 - Draw the structures of the chief product formed...Ch. 13 - Draw the structures of the chief product formed...Ch. 13 - Draw the structures of the ethers that can be...Ch. 13 - Prob. 13.25ECh. 13 - Give the structure of an alcohol that could be...Ch. 13 - Give the structure of an alcohol that could be...Ch. 13 - What products would result from the following...Ch. 13 - What products would result from the following...Ch. 13 - Each of the following conversions requires more...Ch. 13 - Each of the following conversions requires more...Ch. 13 - The three-carbon diol used in antifreeze is It is...Ch. 13 - Methanol is fairly volatile and evaporates quickly...Ch. 13 - Prob. 13.34ECh. 13 - Prob. 13.35ECh. 13 - Name an alcohol used in each of the following...Ch. 13 - Prob. 13.37ECh. 13 - Prob. 13.38ECh. 13 - Assign a common name to each of the following...Ch. 13 - Assign a common name to each of the following...Ch. 13 - Assign the IUPAC name to each of the following...Ch. 13 - Assign the IUPAC name to each of the following...Ch. 13 - Prob. 13.43ECh. 13 - Draw structural formulas for the following: a....Ch. 13 - Prob. 13.45ECh. 13 - Prob. 13.46ECh. 13 - Prob. 13.47ECh. 13 - Arrange the following compounds in order of...Ch. 13 - Arrange the compounds in Exercise 13.48 in order...Ch. 13 - Prob. 13.50ECh. 13 - Complete the following reactions: a. b....Ch. 13 - Prob. 13.52ECh. 13 - Lipoic acid is required by many microorganisms for...Ch. 13 - Alcohols and thiols can both be oxidized in a...Ch. 13 - Prob. 13.55ECh. 13 - Prob. 13.56ECh. 13 - Prob. 13.57ECh. 13 - Thiols have lower boiling points and are less...Ch. 13 - Prob. 13.59ECh. 13 - Prob. 13.60ECh. 13 - Prob. 13.61ECh. 13 - Prob. 13.62ECh. 13 - A mixture of ethanol and 1propanol is heated to...Ch. 13 - Prob. 13.64ECh. 13 - Prob. 13.65ECh. 13 - Prob. 13.66ECh. 13 - Prob. 13.67ECh. 13 - Figure 13.8 points out that methanol is used as a...Ch. 13 - Figure 13.13 focuses on the use of thiol chemistry...Ch. 13 - Prob. 13.70ECh. 13 - Prob. 13.71ECh. 13 - Prob. 13.72ECh. 13 - The compound that has the greatest polarity is: a....Ch. 13 - Alcoholic beverages contain: a. wood alcohol. b....Ch. 13 - Prob. 13.75ECh. 13 - Which of the following compounds is an ether? a....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What functional group distinguishes each of the following hydrocarbon derivatives? a. halohydrocarbons b. alcohols c. ethers d. aldehydes e. ketones f. carboxylic acids g. esters h. amines Give examples of each functional group. What prefix or suffix is used to name each functional group? What are the bond angles in each? Describe the bonding in each functional group. What is the difference between a primary, secondary, and tertiary alcohol? For the functional groups in ah, when is a number required to indicate the position of the functional group? Carboxylic acids are often written as RCOOH. What does COOH indicate and what does R indicate? Aldehydes are sometimes written as RCHO. What does CHO indicate?arrow_forward7 8 9arrow_forward8. Name each alkyne. A. CH3CH2CH2C=CH B. CH3CH2CH2C=CCH3arrow_forward
- Which is an example of an ether?A. CH3OH B. CH3CH2CH2Cl C. CH3CH2COOCH3 D. CH3CH2OCH2CH3arrow_forward1. Which functional group is found in aldehydes? a. CHO b. -CH;OH c. -COOH d. RCOR' 2. Which functional group is found in ketones? a. -CHO CH:OH c. -COOH RCOR' 3. Which structural feature is common to aldehydes and ketones? a, an oxygen atom bonded to both a carbon atom and a hydrogen atom b. an oxygen atom bonded to two carbon atoms c. an oxygen atom double bonded to a carbon atom d. two oxygen atoms bonded to the same carbon atom 4. What is the IUPAC name for the following compound? a. I-pentaldehyde b. l-pentanal c. pentanal d. pentanealdehyde Cetoa of Fiarrow_forwardAlkenes can be synthesized from alkyl halides when this compound is used as a reactant:A. Sodium bicarbonateB. Sulfuric acidC. Sodium hydroxideD. Acetic Acidarrow_forward
- Which of the following compounds is methyl formate? a. H3C-C-H mco-on, b. H3C-C- 0-CH3 H3C-C-OH d. -0-CH3 e. H3C-O-c-H What is the IUPAC name for the following compound? Br H3C-CH-CH2-CH a. 2-bromobutanal b. a-bromobutanal c. 3-bromobutanal d. B-bromobutyraldehyde e. 3-bromobutanaldehyde What is the IUPAC name for the following compound? CH3 -CH-CH,-CH-CI CH3 H3C- a. 5-chloro-3-methylhexanone b. 1-chloro-1,3-dimethyl-4-pentanone c. 5-chloro-3,5-dimethyl-2-pentanone d. 5-chloro-3-methyl-2-hexanone e. 2-chloro-4-methyl-5-hexanonearrow_forwardConsider the structure of cyclohexene, if it undergoes epoxidation followed by exposure to water, which of the following final product is formed? a. Cyclohexan-1,2,-diol b. Cyclohexane c. Cyclohexanone d. Hexan-1,-6-dioic acidarrow_forwardc. CH2-CH2-CH2-CH-CH3 b. C CH3 CH3 d. CH3-CH2-CH-CH,-CH--CH3 CH3 111s C. CH3 12.36 Give the IUPAC name for each of the follov ing d. alkanes. a. CH3-CH,--CH-CH2-CH,-CH3 ČH3 12.39 b. CH3--CH2-CH2-CH-CH3 ČH3 CH c. CH-CH2-CH-CH3 ČH3 CH3 d. CH3-CH-CH2-CH-CH3 CH3 CH3 14.4 12.37 Give the IUPAC name for each of the followi ng alkanes. a. CH3-CH-CH2-CH-CH-CH3 CH3 CH3 CH3 CH3 b. CH3--C-CH,-CH-CH3 CH3 CH3 12 CH3 c. CH3-CH2-C-CH2-CH3 CH2 CH3 CH3 d. CH3-CH2-C-CH2-CH2 CH3 CH2 1. CH3 12.38 Give the IUPAC name for each of the follo ng alkanes. a. CH3-CH2-CH-CH-CH-CH-C CH3 CH2 CH3 CH3arrow_forward
- Consider the structure of cyclohexene, if it undergoes strong oxidation, which of the following final product is formed? a. Cyclohexan-1,2,-diol b. Cyclohexane c. Cyclohexanone d. Hexan-1,-6-dioic acidarrow_forwardConsider the following starting material and choose all of the functional groups that are likely to oxidize in it. a. aldehyde b. secondary alcohol c. alkane d. hemiacetal e. carboxylic acid f. alkenearrow_forward4- Choose the correct reactants required to synthesize the following molecule. H3CO OCH 3 A. Acetone and methanal B. Acetone and methanol C. Formaldehyde and propanal D. Formaldehyde and propanol 5- What is the IUPAC name of the following compound? CI A. 6-Chloro-4-ethoxy-1-octanal B. 6-Chloro-4-ethoxyoctanal C. 6-Chloro-4-ethoxycyclooctanal D. 4-Ethoxy-6-chlorooctanal - What is the IUPAC name of the following compound? 0 Br. Br Cl A. 2,6-Dibromo-3-chloro-5,5-dimethylhexanone B. 2,6-Dibromo-3-chloro-5,5-dimethylcyclohexanone C. 2,6-Dibromo-5-chloro-3,3-dimethylhexanone D. 2,6-Dibromo-5-chloro-3,3-dimethylcyclohexanonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning  ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License