
Concept explainers
(a)
Interpretation:
The product should be identified when 1-ethylcyclohexene treated with
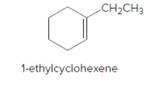
Concept Introduction:
Cyclic
Reaction of an alkene with
Answer to Problem 13.63P
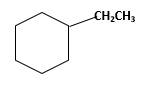
Explanation of Solution
Unsaturated 1-ethylcyclohexene,alkene molecule reacts with
(b)
Interpretation:
The product should be identified when 1-ethylcyclohexene treated with
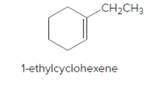
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist of a carbon-carbon double bond which has the general formula of
Reaction of an alkene with
Answer to Problem 13.63P
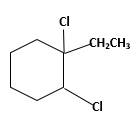
Explanation of Solution
Unsaturated 1-ethylcyclohexene, alkene molecule reacts with
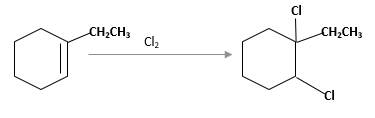
(c)
Interpretation:
The product should be identified when 1-ethylcyclohexene treated with
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist of a carbon-carbon double bond which has the general formula of
Reaction of an alkene with
Answer to Problem 13.63P
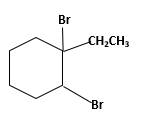
Explanation of Solution
Unsaturated 1-ethylcyclohexene, alkene molecule reacts with
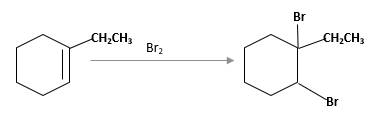
(d)
Interpretation:
The product should be identified when 1-ethylcyclohexane treated with
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist of a carbon-carbon double bond which has the general formula of
Reaction of an alkene with
Hydro halogenation reaction of alkenes follows the Markovnikov's rule.
Answer to Problem 13.63P
Explanation of Solution
Unsaturated 1-ethylcyclohexene, alkene molecule reacts with
Hydro halogenation reaction of alkenes follows the Markovnikov's rule. When
Refer to the below reaction:
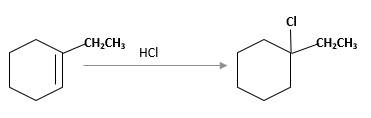
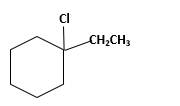
(e)
Interpretation:
The product should be identified when 1-ethylcyclohexene treated with
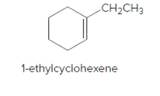
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist of a carbon-carbon double bond which has the general formula of
Reaction of an alkene with
Hydro halogenation reaction of alkenes follows the Markovnikov's rule.
Answer to Problem 13.63P
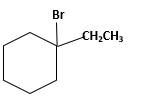
Explanation of Solution
Unsaturated 1-ethylcyclohexene, alkene molecule reacts with
Hydro halogenation reaction of alkenes follows the Markovnikov's rule. When
Refer to the below reaction;
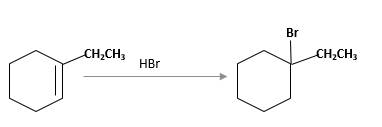
(f)
Interpretation:
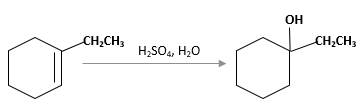
The product should be identified when 1-ethylcyclohexene treated with
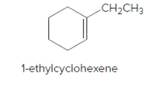
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist of a carbon-carbon double bond which has the general formula of
Reaction of an alkene with
Hydration reaction of alkenes follows the Markovnikov's rule.
Answer to Problem 13.63P
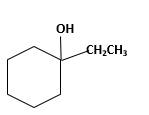
Explanation of Solution
Unsaturated 1-ethylcyclohexene, alkene molecule reacts with
Want to see more full solutions like this?
Chapter 13 Solutions
ALEKS 360 ACCESS CARD F/GEN. ORG.CHEM
- Polyisobutylene, a synthetic high molecular weight alkane with many properties similar to polyethylene (Section 11.5A), is the principal component of Elastol, a product used to clean up oil spills. Elastol dissolves oil to form a thick mat that can be skimmed or suctioned from the surface. Explain why oil dissolves in Elastol.arrow_forwardDraw the structure of each compound. ) p-bromostyrenearrow_forwardHow to convert propene to propanal?arrow_forward
- Draw the neutral organic starting material that will react with Br₂ to form 2-(bromomethyl)tetrahydrofuran. Draw the neutral organic starting material. [= Br₂ CH₂Cl₂arrow_forward● Draw the structures for the three major alkene products that result when 1-pentene is treated with Grubbs catalyst.arrow_forward2. Complete the following reactions for the preparation of alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy CH=CH-CHy + HyO Hydration of alkenes b) Cat. CHy-CHy + H2 drogenation carbonyl pups d)arrow_forward
- What is the structure of the IUPAC name? Draw the structure and give how many carbons are on the parent chain. (2Z)-3-sulfanylhex-2-en-4-ynedialarrow_forwardb). Use ethanol as the only ORGANIC carbon cource, along with any needed reagents to make 4-methylhexan-3-one 4-methylhexan-3-onearrow_forwardDraw the structures of the missing reagent, reactant, or major organic productsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
