
Concept explainers
(a)
Interpretation:
The reactant that has to be used to prepare the given compound from cyclopentene has to be given.
Concept Introduction:
In this reaction no atoms or group of atoms are removed. Instead the unsaturated bond is reduced to saturated bond. A general scheme for addition reaction of
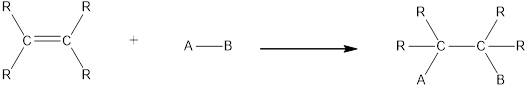
Hydrogenation is an example of addition reaction. In this reaction, a hydrogen molecule is incorporated into the molecules of organic compound. Hydrogenation of alkene results in the formation of alcohol, where both carbon atoms bonded by double bond gets hydrogen atom. This reaction requires a metal as catalyst.
(b)
Interpretation:
The reactant that has to be used to prepare the given compound from cyclopentene has to be given.
Concept Introduction:
Chemical reaction in which an atom or a group of atoms are added to each carbon atom of a carbon‑carbon multiple bond in a hydrocarbon or hydrocarbon derivative is known as addition reaction.
In this reaction no atoms or group of atoms are removed. Instead the unsaturated bond is reduced to saturated bond. A general scheme for addition reaction of alkene can be given as shown below,
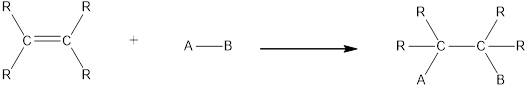
Hydration is an example of addition reaction. In this reaction, a water molecule is incorporated into the molecules of organic compound. Hydration of alkene results in the formation of alcohol, where one carbon atom gets hydrogen atom added and the other carbon atom gets hydroxyl group added to it. This reaction requires a small amount of sulphuric acid as catalyst.
(c)
Interpretation:
The reactant that has to be used to prepare the given compound from cyclopentene has to be given.
Concept Introduction:
Chemical reaction in which an atom or a group of atoms are added to each carbon atom of a carbon‑carbon multiple bond in a hydrocarbon or hydrocarbon derivative is known as addition reaction.
In this reaction no atoms or group of atoms are removed. Instead the unsaturated bond is reduced to saturated bond. A general scheme for addition reaction of alkene can be given as shown below,
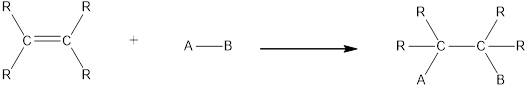
Halogenation is an example of addition reaction. In this reaction, a halogen molecule is incorporated into the molecules of organic compound. Halogenation of alkene results in the formation of dihaloalkane, where both carbon atoms bonded by double bond gets halogen atom.
(d)
Interpretation:
The reactant that has to be used to prepare the given compound from cyclopentene has to be given.
Concept Introduction:
Chemical reaction in which an atom or a group of atoms are added to each carbon atom of a carbon‑carbon multiple bond in a hydrocarbon or hydrocarbon derivative is known as addition reaction.
In this reaction no atoms or group of atoms are removed. Instead the unsaturated bond is reduced to saturated bond. A general scheme for addition reaction of alkene can be given as shown below,
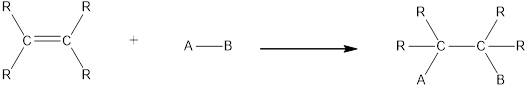
Hydrohalogenation is an example of addition reaction. In this reaction, a hydrogen halide molecule is incorporated into the molecules of organic compound. Hydrohalogenation of alkene results in the formation of
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
GENERAL,ORGANIC,+BIO.CHEM.-MINDTAP
- Supply the structural formula of the product in each of the following alkene addition reactions. a. CH3CH=CHCH3 + Cl2 ? b. CH3CHCH=CH2 + HCl ?arrow_forwardDraw a condensed structural formula for each of the following unsaturated hydrocarbons. a. Propylene b. Methylenecyclopentane c. Vinyl iodide d. Allyl chloridearrow_forwardGive the name for the product from the hydrogenation of each of the following. 3-methyl-2-hexene cyclopentenearrow_forward
- Draw the correct structure for the product resulting from the addition of hydrogen gas to trans-3-hexene.arrow_forwardThe correct synthesis of octane would involve which of the following reactions?arrow_forwardWhich type of reaction will an alkene not undergo? Ohydrogenation halogenation oxidation dehydration polymerizationarrow_forward
- In general, what type of compound is expected to be the final product when two halogen atoms add across the double bond of an alkene?arrow_forwardExplain by the equation how cyclohexane can be obtained from benzene?arrow_forwardwhat type of reaction will give the following conversion: Ethanol Ethene Substitution Reaction Condensation Reaction Elimination Reaction Addition Reactionarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



