
Concept explainers
Freezing temperature. Ethylene glycol and propylene glycol are liquids used in antifreeze and deicing solutions. Ethylene glycol is listed as a hazardous chemical by the Environmental Protection Agency, while propylene glycol is generally regarded as safe. Table 11 lists the freezing temperature for various concentrations (as a percentage of total weight) of each chemical in a solution used to deice airplanes. A linear regression model for the ethylene glycol data in Table 11 is
where
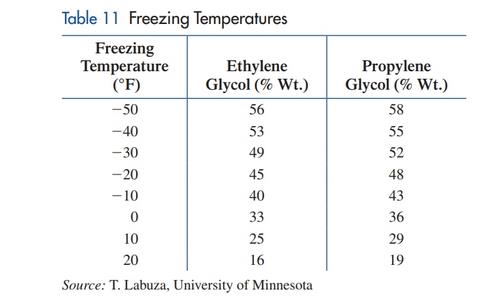
(A) Draw a
(B) Use the model to estimate the freezing temperature to the nearest degree of a solution that
is 30% ethylene glycol.
(C) Use the model to estimate the percentage of ethylene glycol in a solution that freezes at
15°F.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Finite Mathematics for Business, Economics, Life Sciences and Social Sciences
Additional Math Textbook Solutions
Calculus for Business, Economics, Life Sciences, and Social Sciences (13th Edition)
Fundamentals of Differential Equations and Boundary Value Problems
Excursions in Modern Mathematics (9th Edition)
Calculus Volume 2
Thinking Mathematically (6th Edition)
Using and Understanding Mathematics: A Quantitative Reasoning Approach (6th Edition)
 Elementary Linear Algebra (MindTap Course List)AlgebraISBN:9781305658004Author:Ron LarsonPublisher:Cengage Learning
Elementary Linear Algebra (MindTap Course List)AlgebraISBN:9781305658004Author:Ron LarsonPublisher:Cengage Learning Algebra and Trigonometry (MindTap Course List)AlgebraISBN:9781305071742Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage Learning
Algebra and Trigonometry (MindTap Course List)AlgebraISBN:9781305071742Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage Learning Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning


