
Concept explainers
Define each of the following:
a.
b. Brønsted—Lowry acid
c. Lewis acid
Which of the definitions is most general? Write reactions to justify your answer.
(a)
Interpretation: The definition of each term, Arrhenius acid, Bronsted-Lowry acid and Lewis acid is to be given. The most general definition from these terms is to be identified and the reactions are to be given for the justification of answers.
Concept introduction: An acid is a substance that turns litmus to red. It has a sour taste.
Answer to Problem 1RQ
Answer
Arrhenius acid produces hydrogen ions in aqueous solution.
Explanation of Solution
To define: Arrhenius acid.
The substance that produces hydrogen ions in aqueous solution is known as Arrhenius acid.
Arrhenius postulated the concept of acid. According to him “The substance that produces hydrogen ions in aqueous solution is known as acid.”
(b)
Interpretation: The definition of each term, Arrhenius acid, Bronsted-Lowry acid and Lewis acid is to be given. The most general definition from these terms is to be identified and the reactions are to be given for the justification of answers.
Concept introduction: An acid is a substance that turns litmus to red. It has a sour taste.
Answer to Problem 1RQ
Answer
Bronsted-Lowry acids are able to donate a proton.
Explanation of Solution
To define: Bronsted-Lowry acid.
The substance that can donate a proton
Johannes Bronsted and Thomas Lowry proposed a model known as Bronsted-Lowry model. According to this model “The donor of proton
(c)
Interpretation: The definition of each term, Arrhenius acid, Bronsted-Lowry acid and Lewis acid is to be given. The most general definition from these terms is to be identified and the reactions are to be given for the justification of answers.
Concept introduction: An acid is a substance that turns litmus to red. It has a sour taste.
Answer to Problem 1RQ
Answer
Lewis acids can accept a pair of electrons.
The most general definition of acid is given by Bronsted-Lowry model.
Explanation of Solution
To define: Lewis acid.
The chemical species that accepts a pair of non-bonding electrons is known as Lewis acid.
The Gilbert Newton Lewis suggested acid-base theory. According to this theory “The species that can accept a pair of non bonding electrons is known as acid.”
The most general definition of acid is given by Bronsted-Lowry model because this model is applicable to the non-aqueous solution also.
Arrhenius acid,
The substance that produces hydrogen ions in aqueous solution is known as Arrhenius acid.
The dissociation reaction of
The compound
Bronsted-Lowry acid,
The donor of proton
The reaction of
Lowry acid,
The chemical compound that accepts a pair of non-bonding electrons is known as Lewis acid.
The compound
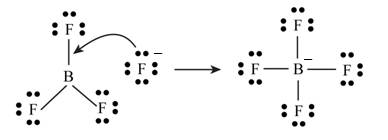
Figure 1
Want to see more full solutions like this?
Chapter 13 Solutions
Bundle: Chemistry: An Atoms First Approach, 2nd, Loose-Leaf + OWLv2, 4 terms (24 months) Printed Access Card
- Define each of the following: a. Arrhenius acid b. BronstedLowry acid c. Lewis acid Which of the definitions is most general? Write reactions to justify your answer.arrow_forwardClassify each of the following statements as true or false: aAll Brnsted-Lowry acids are Arrhenius acids. bAll Arrhenius bases are Brnsted-Lowry bases, but not all Brnsted-Lowry bases are Arrhenius bases. c HCO3 is capable of being amphoteric. d HS is the conjugate base of S2. eIf the species on the right side of an ionization equilibrium are present in greater abundance than those on the left, the equilibrium is favored in the forward direction. f NH4+ cannot act as a Lewis base. gWeak bases have a weak attraction for protons. hThe stronger acid and the stronger base are always on the same side of a proton transfer reaction equation. iA proton transfer reaction is always favored in the direction that yields the stronger acid. jA solution with pH=9 is more acidic than one with pH=4. kA solution with pH=3 is twice as acidic as one with pH=6. lA pOH of 4.65 expresses the hydroxide ion concentration of a solution in three significant figures.arrow_forwarda Write a net ionic equation to show that hypochlorous acid behaves as a Brnsted-Lowry acid in water. b The substance triethanolamine is a weak nitrogen-containing base, like ammonia. Write a net ionic equation to show that triethanolamine, C6H15O3N, behaves as a Brnsted-Lowry base in water.arrow_forward
- Define each of the following: a. Arrhenius acid b. BronstedLowry acid c. Lewis acid Which of the definitions is most general? Write reactions to justify your answer.arrow_forwardList several common acids and where they might be found.arrow_forwardWrite a brief description of the relationships among each of the following groups of terms or phrases. Answers to the Concept-Linking Exercises are given at the end of the chapter. Terms from optional sections 17.1 and 17.3 are in italics. Exercise 11 covers terms related to logarithms, and it is also optional. Arrhenius acid, Brnsted-Lowry acid, Lewis acidarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





