
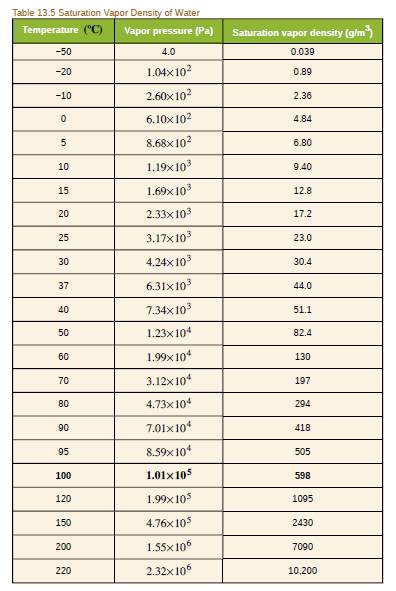
Because humidity depends only on water's vapor pressure and temperature, are the saturation vapor densities listed in Table 13.5 valid in an atmosphere of helium at a pressure of
Trending nowThis is a popular solution!

Chapter 13 Solutions
COLLEGE PHYSICS
Additional Science Textbook Solutions
Tutorials in Introductory Physics
Sears And Zemansky's University Physics With Modern Physics
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
University Physics Volume 2
University Physics with Modern Physics (14th Edition)
Conceptual Physics (12th Edition)
- A deep-sea diver should breathe a gas mixture that has the same oxygen partial pressure as at sea level, where dry air contains 20.9% oxygen and has a total pressure of 1.01×105 N/m2 . (a) What is the partial pressure of oxygen at sea level?(b) If the diver breathes a gas mixture at a pressure of 2.00×106 N/m2 , what percent oxygen should it be to have the same oxygen partial pressure as at sea level?arrow_forwardDetermine at what altitude in meters the air pressure is twice less than on the sea level using the barometic equation that is pressure as a function of height. Given that the temperature is 288.15 K. P=P0e -Mgh/RT, where Po is the pressure at sea level, M is the molar mass of air (in kg/mol), T is standard temperature, R is universal gas constant (8.314 N.m/mol K).arrow_forwardPlease asaparrow_forward
- The table given shows the vapor pressure of water at 20.0ºC as 2.33×103 Pa. Use the ideal gas law to calculate the density of water vapor in g /m3 that would create a partial pressure equal to this vapor pressure. Compare the result with the saturation vapor density given in the table.arrow_forwardAssuming air as the perfect gas, find the density of air at an altitude of 10000 feet at which the pressure is 50225 Pa, assuming the International Standard Atmosphere (ISA) weather conditions at sea level. (Under standard atmospheric conditions, the air pressure is 101325 Pa, its density is 1,225 kg / m3, its temperature is 15 0C. In every 1000 feet in the troposphere, the temperature drops by 2 K.)arrow_forwardThe mercury level in the left arm of the J-shaped tube is attached to a thermostat gas-containing bulb. The left arm is 10.83 cm and the right arm is 34.71 cm above the bottom of the manometer. If the barometric pressure reads 738.4 Torr, what is the pressure of the gas? Assume that temperature-induced changes in the reading of the barometer and J tube are small enough to neglect. Include a drawing of the diagramarrow_forward
- The pressure in an automobile tire depends on the tem- perature of the air in the tire. When the air temperature is 25°C, the pressure gage reads 210 kPa. If the volume of the tire is 0.025 m³, determine the pressure rise in the tire when the air temperature in the tire rises to 50°C. Also, determine the amount of air that must be bled off to restore pressure to its original value at this temperature. Assume the atmospheric pressure to be 100 kPa. Question 2 V= 0.025 m3 T= 25YC P= 210 kPa AIRarrow_forwardOn a day when atmospheric pressure is 76 cmHg, the pressure gauge on a tank 4. On a day when atmospheric pressure is 76 cmHg, the pressure gauge on a tank reads the pressure inside to be 400 cmHg. The gas in the tank has a temperature reads the pressure inside to be 400 cmHg. The gas in the tank has a temperature of 9°C. If the tank is heated to 31°C by the Sun, and if no gas exits from it, what of 9°C. If the tank is heated to 31°C by the Sun, and if no gas exits from it, what will the pressure gauge read?arrow_forwardCalculate the correction that needs to be applied to a T = 89°C if barometric pressure when measuring the boiling point of water is 4.4 mm below the 760mm on a Hg column. Hint: An atmospheric pressure change that increases the height of the column of mercury will increase the boiling point by 0.037° C (0.067° F) for each 1.0mm of additional height.arrow_forward
- Consider a hypothetical atmosphere consisting only of helium (mass = 4 amu) and argon (m = 40 amu). At ground level, the density ratio of the two gases is nHe/ nAr 3.0\times 10-4. At an altitude of 155 km, the density ratio is found to be nHe/nAr = 1.0\times 10-3. Assume the transition from a "well mixed" homosphere to the heterosphere occurs abruptly at the turbopause, which is located somewhere below 155 km Find the altitude where this transition occurs; report your answer in km to 2 significant digits. Assume an isothermal temperature of 1500 K and a constant gravitational acceleration of g = 9.5 m/s2.arrow_forwardA deep-sea diver should breathe a gas mixture that has the same oxygen partial pressure as at sea level, where dry air contains 20.9% oxygen and has a total pressure of 1.01 X 105 N/m2. (a) What is the partial pressure of oxygen at sea level? (b) If the diver breathes a gas mixture at a pressure of 2.00 X 106 N/m2, what percent oxygen should it be to have the same oxygen partial pressure as at sea level?arrow_forwardOn Mars, the atmosphere is composed mainly of carbon dioxide. The value of the gas constant for the Martian atmosphere is 192 J/kg-K, and the acceleration of gravity there is 3.72 m/s?. At the average level of the Martian surface, the average temperature is 230 K, the pressure is 790 N/m?. At a certain altitude, the pressure is 680 N/m? and the average temperature is 203 K. 6. The temperature lapse rate is: A) – 0.01613 K/m B) – 0.1613 K/m C) – 1.613 K/m D) None of the above 7. The altitude is: A) 1200 m B) 1675 m C) 1765 m D) None of the abovearrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





