
Concept explainers
(a)
Interpretation:
The
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bond strength is depends on the formation of the radical, if the radical is involving in resonance which is weakest bond strength.
Chlorination:
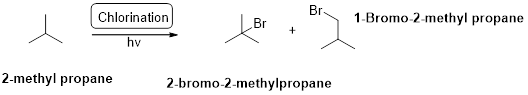
2-methyl propane undergoes radical chlorination which yields the 2-bromo-2-methylpropane and 1-bromo-2-methyl propane.
(b)
Interpretation:
The alkane with molecular formula,
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bond strength is depends on the formation of the radical, if the radical is involving in resonance which is weakest bond strength.
Chlorination:
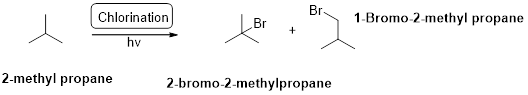
2-methyl propane undergoes radical chlorination which yields the 2-bromo-2-methylpropane and 1-bromo-2-methyl propane.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic Chemistry & Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Consider the tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each six-membered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.arrow_forwardWhat alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2?arrow_forward1. What is the alkane product from the reaction of C7H13COONa and NaOH? a. pentane b. hexane c. butane d. heptane 2. What is the resulting alkane if we have C5H11F and a C5H11F as reactants in Wurtz synthesis? a. hexane b. octane c. nonane d. decane 3.What is the resulting alkane if we have 2C2H5Cl as reactants in Wurtz Synthesis reaction? a. ethane b. butane c. hexane d. octanearrow_forward
- if the heat of hydrogenation for cyclohexene is 120 kj mol and 1,3 cyclohexane is less than 240 kj mol, what is the heat of hydrogenation for benzene? thank you ^___^arrow_forwardThe same alkane is obtained from the catalytic hydrogenation of both alkene A and alkene B. The heat of hydrogenation of alkene A is 29.8 kcal/mol, and the heat of hydrogenation of alkene B is 31.4 kcal/mol. Which alkene is more stable?arrow_forwardConsider the attached tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each sixmembered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.arrow_forward
- Indicate whether each statement is true or false. (a) Butanecontains carbons that are sp2 hybridized. (b) Cyclohexaneis another name for benzene. (c) The isopropyl group containsthree sp3-hybridized carbons. (d) Olefin is anothername for alkyne.arrow_forward1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?arrow_forward1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.arrow_forward
- a. Which is the most stable: 3,4-dimethyl-2-hexene, 2,3-dimethyl-2-hexene, or 4,5-dimethyl-2-hexene? b. Which compound has the largest heat of hydrogenation? c. Which compound has the smallest heat of hydrogenation?arrow_forwardHow many products of 2-hexene+Cl2 are therearrow_forwardHow many stereoisomers are formed from the reaction of cyclohexene with NBS?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





