
Concept explainers
(a)
Interpretation:
The following conversion should be explained.
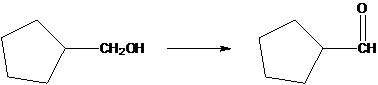
Concept Introduction:
A catalyst is the molecule which is used to speed up the chemical reaction, not being consumed within the procedure. Acids are used as catalysts are needed for an acid catalyzed hydration.
A dehydration reaction is aeration when any organic substance loses the water molecule to form an
(b)
Interpretation:
The following conversion should be explained.

Concept Introduction:
Chemical reaction is the procedure to transform the chemical substance in the new substance. During this procedure sometimes, acid is involved which is the substance which could either accept electron pairs or donate protons in the reactions.
A catalyst is the molecule which is used to speed up the chemical reaction, not being consumed within the procedure. Acids are used as catalysts are needed for an acid catalyzed hydration.
A dehydration reaction is aeration when any organic substance loses the water molecule to form an alkene.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Introduction To General, Organic, And Biochemistry
- 14-70 Show how to prepare each compound from 2-methylcyclohexanol.arrow_forward14-21 Show hydrogen bonding between the oxygen of di ethyl ether and a hydrogen of water.arrow_forward14-57 (Chemical Connections 14E) What are the advantages and disadvantages of using diethyl ether as an anesthetic?arrow_forward
- 17-26 Account for the fact that acetone has a higher boiling point (56°C) than ethyl methyl ether (11°C) even though their molecular weights are almost the same.arrow_forward14-20 Show hydrogen bonding between methanol and water in the following ways. Between the oxygen of methanol and a hydrogen of water Between the hydrogen of methanol’s OH group and the oxygen of waterarrow_forwardExplain the Synthesizing an Ether by a Two-Step Reaction Sequence ?arrow_forward
- How would you convert benzoic acid (C6H5CO2H) to each compound?arrow_forward14-55 (Chemical Connections 140 The legal definition of being under the influence of alcohol is based on blood alcohol content. What is the relationship between breath alcohol content and blood alcohol content?arrow_forward14-64 Explain why the boiling point of ethylene glycol (198°C) is so much higher than that of 1-propanol (97°C), even though their molecular weights are about the same.arrow_forward
- In addition to not undergoing substitution reactions, the alkyl bromide shown in Problem 11-32 also fails to undergo an elimination reaction when treated with base. Explain.arrow_forward14-9 What is the difference in structure between a primary, a secondary, and a tertiary alcohol?arrow_forwardWhat two functional groups react to form the following? a. A hemiacetal b. An acetal c. A ketal d. A hemiketalarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


