
Pearson eText Conceptual Integrated Science -- Instant Access (Pearson+)
3rd Edition
ISBN: 9780135626573
Author: Paul Hewitt, Suzanne Lyons
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 13, Problem 55TE
Use the following illustration to answer exercises 54-56.
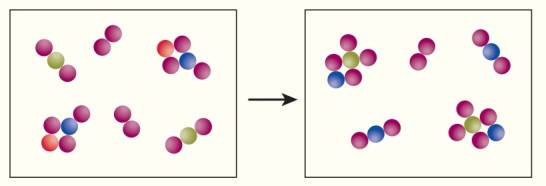
There is an excess of at least one of the reactant molecules. Which one?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Use the data provided for tert-Butyl hydroperoxide, AIBN, and Potassium persulfate.
a) Plot the relationships between kd (s-1) and T (Kelvin) for each compound.
b) Using an Arrhenius fit, what is the activation energy of decomposition for these initiators
c) What is the half-life (in hours) of each compound at the highest temperature data point given
Need part C please, but if all can be done it would be appreciated
Write the general molecular transport equation and identify the functions of each parameters considered in the equation.
:
P
Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products.
In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that
you like, so long as they aren't touching.
H+
H+
+ -OH
☑
Y
Predict the organic products that form in the reaction below:
Click and drag to start drawing a
structure.
Chapter 13 Solutions
Pearson eText Conceptual Integrated Science -- Instant Access (Pearson+)
Ch. 13 - Prob. 1RCCCh. 13 - Prob. 2RCCCh. 13 - Prob. 3RCCCh. 13 - If it takes 436 kilojoules to break a bond, how...Ch. 13 - What is released by an exothermic reaction?Ch. 13 - What is absorbed by an endothermic reaction?Ch. 13 - Prob. 7RCCCh. 13 - What generally happens to the rate of a chemical...Ch. 13 - Which reactant molecules are the first to pass...Ch. 13 - How is an acid different from a base?
Ch. 13 - When an acid is dissolved in water, what ion does...Ch. 13 - Prob. 12RCCCh. 13 - Are there many hydronium ions in neutral water?Ch. 13 - What is true about the relative concentrations of...Ch. 13 - What does the pH of a solution indicate?Ch. 13 - Prob. 16RCCCh. 13 - What elements have the greatest tendency to behave...Ch. 13 - What happens to a reducing agent as it reduces?Ch. 13 - What metal coats a galvanized nail?Ch. 13 - What is iron forced to accept during cathodic...Ch. 13 - What happens to the polarity of oxygen atoms as...Ch. 13 - What catalyst is effective in the destruction of...Ch. 13 - Prob. 23TISCh. 13 - What net effect does a chemical reaction have on a...Ch. 13 - What is the product of the reaction between carbon...Ch. 13 - Prob. 26TISCh. 13 - Prob. 27TISCh. 13 - A material that tends to lose electrons is put...Ch. 13 - What is the primary difference between a battery...Ch. 13 - Prob. 30TISCh. 13 - Rank these reaction profiles in order of...Ch. 13 - Rank the covalent bonds in order of increasing...Ch. 13 - Review the concept of electronegativity in Section...Ch. 13 - Review the concept of electronegativity in Section...Ch. 13 - Rank these molecules from least oxidized to most...Ch. 13 - Prob. 44TSCh. 13 - Prob. 45TSCh. 13 - When the hydronium ion concentration of a solution...Ch. 13 - When the pH of a solution is 1, the concentration...Ch. 13 - Show that the pH of a solution is 0.301 when its...Ch. 13 - Show that the hydroxide ion concentration of a...Ch. 13 - How can 50g of wood burn to produce more than 50g...Ch. 13 - Balance these equations: a Fe(s)+O2(g)Fe2O3(s) b...Ch. 13 - Balance these equations: a Fe(s)+S(s)Fe2S3(s) b...Ch. 13 - Prob. 53TECh. 13 - Use the following illustration to answer exercises...Ch. 13 - Use the following illustration to answer exercises...Ch. 13 - What changes during a chemical reaction?Ch. 13 - Prob. 58TECh. 13 - Is photosynthesis an exothermic or endothermic...Ch. 13 - Why does blowing into a campfire make the fire...Ch. 13 - In the laboratory, endothermic reactions are...Ch. 13 - Prob. 62TECh. 13 - Why does a glowing splint of wood burn only slowly...Ch. 13 - Prob. 64TECh. 13 - Chew a salt-free soda cracker for a few minutes...Ch. 13 - Prob. 66TECh. 13 - Does the ozone pollution from automobiles help...Ch. 13 - Chlorine is put into the atmosphere by volcanoes...Ch. 13 - Prob. 69TECh. 13 - Prob. 70TECh. 13 - An acid and a base react to form salt, which...Ch. 13 - Identify the acid or base behavior of each...Ch. 13 - Prob. 73TECh. 13 - Prob. 74TECh. 13 - The main component of bleach is sodium...Ch. 13 - Prob. 76TECh. 13 - Prob. 77TECh. 13 - Within a neutral solution of supercritical water...Ch. 13 - What is the concentration of hydronium ions in a...Ch. 13 - Can an acidic solution be made less acidic by...Ch. 13 - How does burning fossil fuels lower the pH of the...Ch. 13 - Bubbling carbon dioxide into water causes the pH...Ch. 13 - Pour vinegar onto beach sand from the Caribbean,...Ch. 13 - What happens to the pH of soda water as it loses...Ch. 13 - Prob. 85TECh. 13 - Prob. 86TECh. 13 - Why is the chlorine atom such a strong oxidizing...Ch. 13 - Prob. 88TECh. 13 - What element behaves as the oxidizing agent in the...Ch. 13 - Hydrogen sulfide, H2S, burns in the presence of...Ch. 13 - Unsaturated fatty acids, such as C12H22O2, react...Ch. 13 - The type of iron that the human body needs for...Ch. 13 - Why is lithium a preferred metal for the making of...Ch. 13 - Chemical equations must be balanced not only in...Ch. 13 - Study question 94 before attempting to balance...Ch. 13 - How does turning on the radio while you are...Ch. 13 - What are some key advantages that a fuel-cell...Ch. 13 - Do our bodies gradually oxidize or reduce the food...Ch. 13 - Pennies manufactured after 1982 are made of zinc...Ch. 13 - Water is 88.88 oxygen by mass. Oxygen is exactly...Ch. 13 - Why is the air over an open flame always moist?Ch. 13 - Upon ingestion, grain alcohol, C2H6O, is...Ch. 13 - Prob. 103TDICh. 13 - Can industries be trusted to self-regulate the...Ch. 13 - In the centralized model for generating...Ch. 13 - Prob. 1RATCh. 13 - Prob. 2RATCh. 13 - How much energy, in kilojoules, is released or...Ch. 13 - The yeast in bread dough feeds on sugar to produce...Ch. 13 - What role do CFCs play in the catalytic...Ch. 13 - What is the relationship between the hydroxide ion...Ch. 13 - When the hydronium ion concentration equals 1 mole...Ch. 13 - Prob. 8RATCh. 13 - Why does a battery that has thick zinc walls last...Ch. 13 - What element is oxidized in this equation and what...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Orbital resonances like those among Io, Europa, and Ganymede are the results of extremely rare accidents, so we...
Life in the Universe (4th Edition)
The equivalent capacitance of three capacitors in FIGURE P23.37.
College Physics: A Strategic Approach (3rd Edition)
The force, when you push against a wall with your fingers, they bend.
Conceptual Physics (12th Edition)
20. A copper transmission cable 100 km long and 10.0 cm in diameter carries a current of 125 A. What is the pot...
College Physics (10th Edition)
The range of wavelengths corresponding to UV waves, X-rays, visible light in scientific notations.
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- How do you identify the unknown from simple distillation which was analyzed by a GC? And How the flow rate and column temperature effects on the retention time of a compound on a GC column?arrow_forwardWhat is the standard enthalpy of a reaction for which the equilibrium constant is (i) doubled, (ii) halved when the temperature is increased by 10 K at 298 K?arrow_forwardi) Explain the Hexagonal Closed Packing (HCP) unit cell with a neat diagram and provide a minimum of two examples for HCP structure. ii) Explain Self interstitial atom and Interstitial impurity atom with a neat diagram.arrow_forward
- Your friend refuses to use the coffee creamers at the local diner because they have been sitting on the table all day. Explain to your friend why these creamers are perfectly safe to use.arrow_forwardWhat is the difference in packing efficiency between the hcp structure and the ccp structure?arrow_forwardCompare interstitial and vacancy atomic mechanisms for diffusion and (b) cite two reasons why interstitial diffusion is normally more rapid than vacancy diffusion.arrow_forward
- Define an equilibrium constant for gas-phase reaction.arrow_forwardYou have been tasked with scaling up the capture chromatography step that you designed in lab for the GFPuv process. Your large-scale column will be used to load approximately 100 L of the same clarified lysate. So that you all use the same values in your calculation, assume that you loaded 7.5 CV of clarified lysate to a 45 mL column during development studies. The concentration of GFP in the clarified lysate is 1.89 mg GFP/mL lysate. What is the height of the production column? What diameter of column is required? What volumetric flow rate, in L/h, will you use at the larger scale?arrow_forwardPlease tell me the solution process why the value comes out at low temperature.arrow_forward
- Why we use a number of antimony-bismuth couple in a thermopile?arrow_forward(a) A commercial 737 jet transporting 143 passengersand 5 crew members from Kansas City (MCI) to Baltimore (BWI) burned 11,800 lb (about 1700 gallons)of Jet A fuel en route. Jet A fuel is kerosine based,consisting primarily of CnH2n+2 hydrocarbons, withn: 5 6 to 16, so the carbonhydrogen ratio is close to12. During this flight, how much CO2 was releasedinto the atmosphere? Assume the combustion of thefuel was complete, so all the fuel was burned to formCO2 and H2O. Give both the mass of CO2 produced(in kg and in lbs) and the volume it would occupy at298 K, 1 atm.(b) How much CO2 would be released into the atmosphere if those passengers and crew made the tripinstead, in pairs, in hybrid cars at 40 miles per gallon.Assume the density of the gasoline is 0.75 kg L21andthat carbon and hydrogen dominate the compositionin a ratio of 1:2. The road trip is 1082 miles.arrow_forwardQ1: The following reactions were found to occur while trying to make a desired product B A B -TAI=K₁AC₁ A-X -TA2 = K₂A CA A+X-Y -TA3=K3A CACX Species X and Y are both foul pollutants a) What is the point selectivity of B with respect to the foul pollutants X and Y? Plot the point selectivity as function of CA (CAo=1 M) at 300 K. b) How would you carry out this reaction to maximize the formation of B (type of reactor preferred)? Just give a recommendation based on (a). Additional Information -10,000/T KIA =.5 e min Tin degrees Kelvin -20,000/T K2A = 50 e min T in degrees Kelvin min¹, T in degrees Kelvin K3A = 100 e 5,000/Tarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning

Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning

The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY