
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
4th Edition
ISBN: 9781119234715
Author: Klein
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13, Problem 77CP
Interpretation Introduction
Interpretation: The mechanism for the given reaction needs to be proposed.
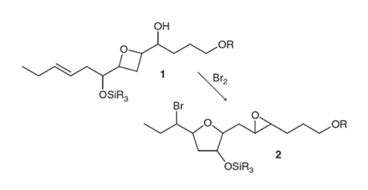
Concept Introduction: When in any chemical situation there are two possible reactions, the product formation depends on the mechanism leading to a stable product. The formation of an intermediate also plays an important role in any reaction. In the given case, electrophilic bromine attacks the given reactant to form a bromonium ion. The formation of an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
help with both parts and step by step mechanism
Answer ALL parts.
(a)
Provide reagents for the following transformation. No mechanism is
required.
NH2
(b)
Suggest structures for A and B, and provide detailed mechanisms for their
formation.
1. CI
он
NaOH (2eq)
A
ACONA/ACOH
B
2. H3O*
heat
(c)
Provide a detailed mechanism for the following transformation and
suggest a chemical structure for C. All reagents are added together and
no additional acid or base is needed.
MENH2
COME
C9H13NO2
lo
(d)
Suggest a retrosynthetic analysis for the following molecule, and outline a
synthetic route, specifying reagents. No mechanisms are required.
3.
vi) please
Chapter 13 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
Ch. 13.2 - Prob. 1LTSCh. 13.2 - Prob. 1PTSCh. 13.2 - Prob. 2PTSCh. 13.2 - Prob. 3ATSCh. 13.4 - Prob. 4CCCh. 13.5 - Prob. 2LTSCh. 13.5 - Prob. 5PTSCh. 13.5 - Prob. 6ATSCh. 13.5 - Prob. 7CCCh. 13.5 - Prob. 8CC
Ch. 13.5 - Prob. 9CCCh. 13.6 - Prob. 10CCCh. 13.7 - Prob. 11CCCh. 13.7 - Prob. 12CCCh. 13.8 - Prob. 3LTSCh. 13.8 - Prob. 13PTSCh. 13.8 - Prob. 14ATSCh. 13.9 - Prob. 15CCCh. 13.10 - Prob. 4LTSCh. 13.10 - Prob. 17ATSCh. 13.10 - Prob. 5LTSCh. 13.10 - Prob. 19ATSCh. 13.11 - Prob. 20CCCh. 13.12 - Prob. 6LTSCh. 13.12 - Prob. 7LTSCh. 13 - Prob. 26PPCh. 13 - Prob. 27PPCh. 13 - Prob. 28PPCh. 13 - Prob. 29PPCh. 13 - Prob. 30PPCh. 13 - Prob. 31PPCh. 13 - Prob. 32PPCh. 13 - Prob. 33PPCh. 13 - Prob. 34PPCh. 13 - Prob. 35PPCh. 13 - Prob. 36PPCh. 13 - Prob. 37PPCh. 13 - Prob. 38PPCh. 13 - Prob. 39PPCh. 13 - Prob. 40PPCh. 13 - Prob. 41PPCh. 13 - Prob. 42PPCh. 13 - Prob. 43PPCh. 13 - Prob. 44PPCh. 13 - Prob. 45PPCh. 13 - Prob. 46ASPCh. 13 - Prob. 47ASPCh. 13 - Prob. 48ASPCh. 13 - Prob. 49ASPCh. 13 - Prob. 50ASPCh. 13 - Prob. 51ASPCh. 13 - Prob. 52ASPCh. 13 - Prob. 53ASPCh. 13 - Prob. 54IPCh. 13 - Prob. 59IPCh. 13 - Prob. 60IPCh. 13 - Prob. 61IPCh. 13 - Prob. 62IPCh. 13 - Prob. 63IPCh. 13 - Prob. 64IPCh. 13 - Prob. 65IPCh. 13 - Prob. 66IPCh. 13 - Prob. 69IPCh. 13 - Prob. 70IPCh. 13 - Prob. 71IPCh. 13 - Prob. 72IPCh. 13 - Prob. 73IPCh. 13 - Prob. 74IPCh. 13 - Prob. 77CPCh. 13 - Prob. 79CPCh. 13 - Prob. 80CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forward
- Draw a structural formula for the major organic product of each reaction and specify the most likely mechanism by which each is formed. (g) CH3CH2ONa++CH2=CHCH2Clethanolarrow_forward(R)-Pulegone, readily available from pennyroyal oil, is an important enantiopure building block for organic syntheses. Propose a mechanism for each step in this transformation of pulegone.arrow_forwardFollowing is the structure of miconazole, the active antifungal agent in a number of over-the-counter preparations, including Monistat, that are used to treat vaginal yeast infections. One of the compounds needed for the synthesis of miconazole is the trichloro derivative of toluene shown on its right. (a) Show how this derivative can be synthesized from toluene. (b) How many stereoisomers are possible for miconazole?arrow_forward
- Propose a mechanism for the following reaction, an important step in the laboratory synthesis of proteins:arrow_forwardWhen cis-2-decalone is dissolved in ether containing a trace of HCl, an equilibrium is established with trans-2-decalone. The latter ketone predominates in the equilibrium mixture. Propose a mechanism for this isomerization and account for the fact that the trans isomer predominates at equilibrium.arrow_forwardgive the mechanism of the following reactions and explain why any selectivities occur.arrow_forward
- Jut L Br HNO3/H₂SO4 NaOEt M N K (i) Give the structure of M and a mechanism for its formation. L and of bromoquinoline Explain the reactivity regioselectivity of the reaction. (ii) Give the structure of N and a mechanism for its formation. and bromoquinoline L Explain the reactivity of regioselectivity of the reaction.arrow_forwardDetailed mechanism of reaction of compound 5 with phosphorous tribromide to give compound 3arrow_forwardThe natural product jasmone can be prepared according to the retrosynthesis outlined below. Jasmone J EtO₂C CO₂Et I Br K F EtO₂C CO₂Et H (i) In the final step alkyne F was converted into jasmone by a partial reduction. Discuss this reaction explaining how the careful choice of reaction conditions can give the chemo- and stereoselectivity required in order to efficiently access jasmone. (ii) Use disconnections to show how cyclopentenone F can be prepared from diketone G. In your answer include clearly site(s) for disconnection and identify synthon structures (mechanisms not required).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY