
Concept explainers
Lakes freeze from top down we all know that ice cubes float in a glass of water. Why? Virtually every substance contracts when it solidifies—the solid is denser than the liquid. If this happened to water. Ice cubes would sink to the bottom of a glass, and ice sheets would sink to the bottom of a lake. Fortunately, this doesn't happen. Liquid water expands by
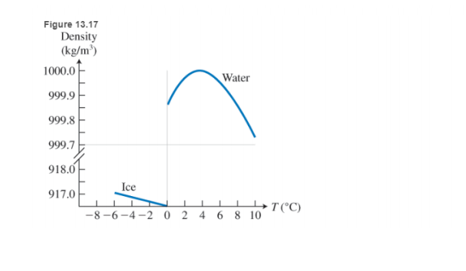
But this is not the only special thing about water. While the density of most substances increases when they are cooled, water density shows a very peculiar temperature dependence (see Figure 13.17). As the temperature decreases, water density increases, but only until
Note that if water were most dense at the freezing point, then in the winter the very cold water at the surface of lakes would sink in this case the lake would freeze from the bottom up, and an life in it would be destroyed.
The expansion of water when it freezes has another important environmental benefit: the so-called freeze-thaw effect on sea memory rocks. Water is absorbed into cracks in these rocks and men freezes in cold weather. The solid ice expands and cracks the rock, like a wood-cutter splitting logs. This continual process of liquid w after absorption, freezing and cracking releases mineral and nitrogen deposits into the soil and can eventually break the rock down into soil.
Why does water freeze from the top down?
a. The denser water at
b. The less dense ice at
c. The denser water at
d. Because of both a and b
e. Because of both b and c
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
COLLEGE PHYSICS:VOL.1
Additional Science Textbook Solutions
College Physics
College Physics: A Strategic Approach (4th Edition)
Essential University Physics (3rd Edition)
Life in the Universe (4th Edition)
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Introduction to Electrodynamics
- Dry air is primarily composed of nitrogen. In a classroom demonstration, a physics instructor pours 2.00 L of liquid nitrogen into a beaker. After the nitrogen evaporates, how much volume does it occupy if its density is equal to that of the dry air at sea level? Liquid nitrogen has a density of 808 kg/m3.arrow_forward1. In Section 4.1 in the description of matter, the following terms were introduced: elements, atoms, electrons, protons, neutrons, nucleus, molecules, mixtures, and solutions. Create a concept map explaining the composition of matter by appropriately organizing and linking these concepts to form meaningful propositions. After completing your concept map, compare your map with that of a classmate or the instructor. Are they same? Should they be? Discuss the similarities and differences that you find between the maps.arrow_forwardBird bones have air pockets in them to reduce their weight—this also gives them an average density significantly less than that of the bones of other animals. Suppose an ornithologist weighs a bird bone in air and in water and finds its mass is 45.0 g and its apparent mass when submerged is 3.60 g (the bone is watertight). (a) What mass of water is displaced? (b) What is the volume of the bone? (c) What is its average density?arrow_forward
- (a) The density of water at 0C is very nearly 1000kg/m3 (it is actually 999.84kg/m3 ), whereas the density of ice at 0C is 917kg/m3. Calculate the pressure necessary to keep ice from expanding when it freezes, neglecting the effect such a large pressure would have on the freezing temperature. (This problem gives you only an indication of how large the forces associated with freezing water might be.) (b) What are the implications of this result for biological cells that are frozen?arrow_forward. The volume of the Drop Tower "Bremen" (a 100-meter-tall tube used to study processes during free fall) is l,700 m3. (a) What is the mass of the air that must he removed from it to reduce the pressure inside to nearly zero (1 Pa compared to 100,000 Pa)? (b) What is the weight of the air in pounds?arrow_forwardLarge helium-filled balloons are used to lift scienti?c equipment to high altitudes. (a) What is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0C and rises to an altitude where its volume is twenty times the original volume and its temperature is 50.0C ? (b) What is the gauge pressure? (Assume atmospheric pressure is constant.)arrow_forward
- An ice cube is placed in a glass of water. What happens to the level of the water as the ice melts?arrow_forward9:02 PM * 84% A webassign.net 14. -/1 POINTS OSCOLPHYS2016 11.5.WA.019. MY NOTES The bottom of a rectangular swimming pool is 11 m × 19 m. If the atmospheric pressure above the swimming pool changes from 743 to 755 mm of mercury, determine the amount by which the force on the bottom of the pool increases? (Assume the density of mercury is 13.6 x 103 kg/m3.) N. Additional Materials Reading Show My Work (Optional) ?arrow_forwardMolecules CO₂ in the liquid phase have many applications for example as fire extinguishers and also materials in food processing. The liquid phase is also the main choice when transporting CO₂ because of its higher density than the gas phase. Below is the phase diagram of CO₂. (1 bar = 1 atmosphere). If we now have a dry ice at a pressure of 1 bar, what should be done so that we can turn it into liquid CO₂ directly? Answer with sufficient arguments 73.8 Solid padat tekanan [bar] 52 -78.5-56.6 Liquid cair gas temperatur [C] 31arrow_forward
- 1. What would happen to the water level in a glass if the ice cube floating in a glass of water will be completely melted? 2. A. Why does a balloon filled with helium gas rise? B. Is there a lower limit on how much helium gas it must contain before it begins to rise?arrow_forwardn 17 Which property of liquids is shown in the picture below? out of 1 question Select one: a. Capillarity b. Cohesion c. Diffusionarrow_forward5.0 L air 2.0L air The volume of a sample of air in a cylinder with a movable piston is 2.0 L at a pressure P1, as shown in the diagram above. The volume is increased to 5.0 L as the temperature is held constant. The pressure of the air in the cylinder is now P2. How does P2 compare to P, for the sample? 2. A. P2 is greater than P1 B. P2 is less than P1 C. P2 is equal to P1 D. It cannot be determined without knowing the temperature of the sample. DELL F6 F7 F8 F9 F2 F3 F4 F5 & @ #3 2$ 8. 3 4 W Y 10 Earrow_forward
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning





