
Interpretation:
The structure of a given molecular formula C11H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C-13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
To Identify:
The structure of an alcohol of given molecular formula C11H6.
Broadband-decoupled spectrum:
The spectrum shows seven signals whereas the given molecular formula also has seven carbon atoms. Thus all the seven carbons have chemically different electronic environments showing signals.
- The signal in the region of 150-220 ppm indicates the carbon atom of carbonyl group (C=O).
- The three signals in the region of 10-25 ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene or methine groups.
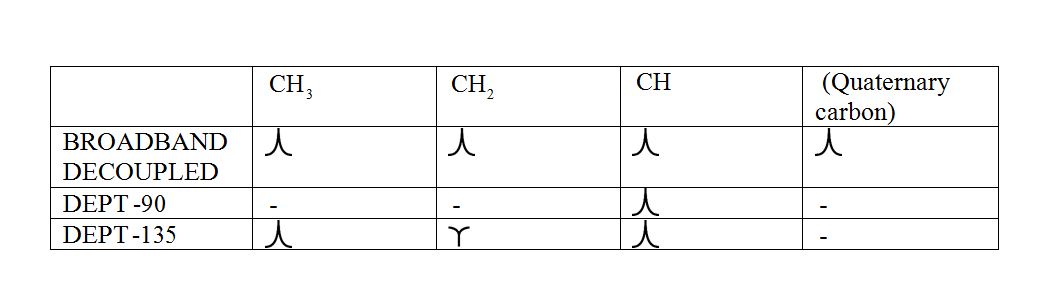
DEPT (Distortionless enhancement by polarization transfer):
a) DEPT-90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
b) DEPT-135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:
Trending nowThis is a popular solution!

Chapter 13 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Propose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm−1 and the following 1H NMR data: Absorption ppm Relative area singlet 1.2 9 triplet 1.3 3 quartet 4.1 2arrow_forwardAddition of m-xylene to the strongly acidic solvent HF/SbF5 at 45C gives a new species, which shows 1H-NMR resonances at 2.88 (3H), 3.00 (3H), 4.67 (2H), 7.93 (1H), 7.83 (1H), and 8.68 (1H). Assign a structure to the species giving this spectrum.arrow_forwardC9H10O2: IR absorption at 1718 cm–1. Propose a structure with data given.arrow_forward
- An organic compound B with formula C6H14O has the following: IR Spectroscopy 2974 cm-1, 1080 cm-1 Mass Spectrometry 102 (M+), 87, 73 1H NMR Spectroscopy Eight signals at δ 1.10 (d, 3H), 1.13 (dd, 3H), 1.14 (dd, 3H), 1.59 (ddq, 1H), 1.60 (ddq, 1H), 3.19 (ddq, 1H), 3.51 (dq, 1H), 3.50 (dq, 1H). Compound B is obtained by the reaction of compound A with NaH followed by CH3CH2Br. The stereochemistry of A is "S" Using this information, deduce a plausible structure for Compound A with correct stereochemistry.arrow_forwardDraw the structure of a compound with the formula C5H10O2 (along with the reasons of choosing it) which, upon analysis, gave key peaks in an infrared spectrum at 3450 cm-1 and 1713 cm-1, as well as the following 1H-NMR spectrum.arrow_forwardGiven the following 13C NMR signals, construct a structure for the unknown compounds. A. Molecular formula: C11H16 Broadband decoupled: 29.5 δ, 31.8 δ, 50.2 δ, 125.5 δ, 127.5 δ, 130.3 δ, 139.8 δ DEPT-90: 125.5 δ, 127.5 δ, 130.3 δ DEPT-135 (positive): 29.5 δ, 125.5 δ, 127.5 δ, 130.3 δ DEPT-135 (negative): 50.2 δ B. The unknown compound has M+∙ = 86 in its mass spectrum and a broad peak at 3400 cm-1 in its IR spectrum. The following are the 13C NMR spectral data: Broadband decoupled: 30.2 δ, 31.9 δ, 61.8 δ, 114.7 δ, 138.4 δ DEPT-90: 138.4 δ DEPT-135 (positive): 138.4 δ DEPT-135 (negative): 30.2 δ, 31.9 δ, 61.8 δ, 114.7 δarrow_forward
- Suggest structures given the 1H NMR spectra and formulas for each of the compounds below. C9H10Oarrow_forward1. There are several isomeric alkanes of molecular formula C6H14.Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (s, 9H), 1.22 (q, 2H) ppmarrow_forwardPropose the 1H and 13C NMR spectrum, including DEPT-90, DEPT-135 and Decoupled, of the following structures.arrow_forward
- Treatment of benzoic acid (C6H5CO2H) with NaOH followed by 1-iodo-3-methylbutane forms H. H has a molecular ion at 192 and IR absorptions at 3064, 3035, 2960–2872, and 1721 cm-1. Propose a structure for H.arrow_forwardAn aromatic compound K, whose molecular formula is C8H11N, is examined in the laboratory to elucidate its structure. The following observations were made: A) Compound K is soluble in dilute hydrochloric acid but insoluble in sodium hydroxide solution. B) Treatment of compound K with excess potassium hydroxide and benzenesulfonyl chloride, C(6)H(5)SO(2)Cl, results in the formation of a heterogeneous mixture. The NMR spectrum of compound K is shown below. C) Compound K when treated with acetic anhydride[CH3-C(O)-O-C(O)-CH3], gives compound L, whose molecular formula is C(10)H(13)ON. Compound L is insoluble in dilute acid or dilute base at room temperature, heating compound L in dilute acid or base, however, regenerates compound K. D) When compound L is heated with a mixture of concentrated nitric acid and sulfuric acid, a single product, compound M, with the molecular formula C(10)H(12)O(3)N(2) is formed in excellent yields. On the basis of these observations draw the structures of…arrow_forwardCompound A is a hydrocarbon with a molar mass of 96g/mol, with the given C13 spectral data. When compound A reacts with BH3 followed by the treatment with basic H2O2 it is converted to compound B. Propose structures for A and B, explain your analysis.Compound A- Proton decoupled C NMR: 26.8, 28.7, 35.7, 106.9, 149.7 δ.DEPT-90: No peak.DEPT-135: No positive peaks; negative peaks at 26.8, 28.7, 35.7, 106.9 δ.Compound B- Proton decoupled C NMR: 26.1, 26.9, 29.9, 40.5, 68.2 δ.DEPT-90: 40.5 δ.DEPT-135: positive peak at 40.5 δ; negative peaks at 26.1, 26.9, 29.9, 68.2 δarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
