
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.13, Problem 13.29P
A bottle of allyl bromide was found to contain a large amount of an impurity. A careful distillation separated the impurity, which has the molecular formula C3H6O. The folowing 13C NMR spectrum of the impurity was obtained:
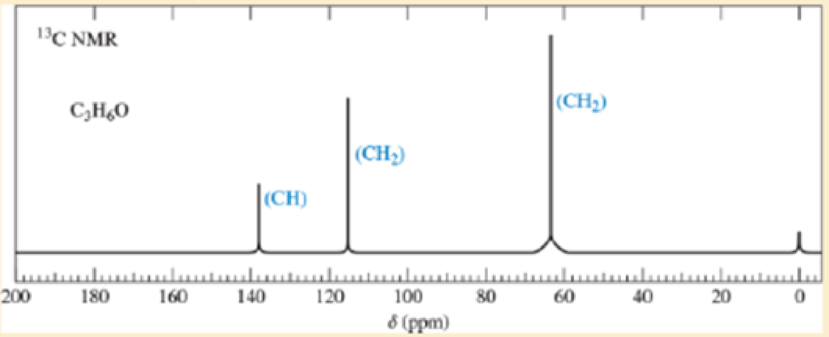
- a. Propose a structure for this impurity.
- b. Assign the peaks in the 13C NMR spectrum to the carbon atoms in the structure.
- c. Suggest how this impunty arose in the allyl bromide sample.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. How many peaks do you expect in the 1³C NMR spectrum for the structure shown?
a. 4
b. 6
OH
c. 7
d. 8
2. How many peaks do you expect in the ¹H NMR spectrum for the structure shown? (
a. 4
b. 6
c. 7
d. 8
H
OH
what compound has this given IR Spectra?
A.
butyraldehyde
B.
butanoic acid
C.
butanone
D.
butanol
30. A compound (C7H₁40) has a strong IR absorption at 1715cm¹. The
¹HNMR spectrum has two signals at d 1.10 (doublet) and d 2.77
(multiplet), ratio 6:1. The 13CNMR spectrum shows three lines at d 218,
39, and 18. Which one of the following structures best fits with this
spectroscopic data? Not.e: d means delta
C
B
A
D
О С
CHO
Chapter 13 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 10. Kuinka monta signaalia seuraava yhdiste antaa irtautetussa ¹3C-NMR-spektrissä? / How many signals does the compound below give in a decoupled ¹3C-NMR spectrum? A) 1 B) 2 C) 3 D) 4arrow_forwardFind how many peaks will appear in the proton (1H) NMR spectrum and carbon (13C) NMR spectrum of each molecue. Don't take into account splitting in the proton(1H) NMRarrow_forwardHow many peaks would you expect to see in the 13C NMR spectrum of this substance? HO HN-N N N OA. 24 OB. 8 OC. 19 D. 21arrow_forward
- 13C NMR is a technique in which the total number of signals represents the number of unique carbon atoms in a molecule. Propose a structure that is consistent with the following data. a. The IR includes peaks at 1603 and 1495 cm-1. b. The 13C NMR has a total of 7 signals. c. The compound has one acidic proton.arrow_forward8 The ¹⁹F nmr spectrum of each of the following molecules exhibits one signal. For which species is this observation consistent with a static molecular structure as predicted by the VSEPR model: a. SiF4 b. PF5 c. SF6 d. SOF2 e. CF4arrow_forward3 2 3 4 2 O (ppm) what structure goes along with this NMR spectra? ONarrow_forward
- How many peaks would each compound show in their 13C NMR spectrum? a) eucalyptol b) linaloolarrow_forwardGiven below are the ¹H NMR spectra of two isomeric compounds with the molecular formula C4H₂O₂. Spectrum X 3 C₂HgO₂ 11 Spectrum Y 10 9 8 0 11 10 ppm Using the following choices, determine which compound corresponds to which spectrum. LOCH 3 A. B. C. b D. C₂H₂O₂ مله 6 OH pom 3 6arrow_forwardThe NMR spectra for compound 1 were acquired in a 7.5 mg / 0.6 mL solution ofCDCl3 . The 1H and 13C peaks are also listedbelow. Provide a full analysis of the NMR spectra for compound 1. correct assignment of NMR spectra of both 13C spectra. correct rationalisation of 13C spectrum1H NMR (400 MHz, CDCl3) δ 7.73 (d, J = 9.5 Hz, 1H), 7.56 (ddd, J = 8.5, 7.5, 1.6 Hz, 1H),7.51 (dd, J = 7.5, 1.6 Hz, 1H), 7.36 (d, J = 8.5 Hz, 1H), 7.30 (dd, J = 8.5, 7.5 Hz, 1H), 6.45(d, J = 9.5 Hz, 1H).13C NMR (101 MHz, CDCl3) δ 160.79, 154.09, 143.43, 131.85, 127.87, 124.44, 118.86,116.94, 116.74.Note: There are two carbon peaks in the 13C spectrum that are so close together that they are not differentiable at the resolution in this experiment. you should be able to assign these peaks to one of two carbon atoms in 1.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY