
(a)
Interpretation:
The product of the hydrogenation reaction of following
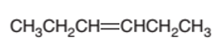
Concept Introduction:
A
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form
(b)
Interpretation:
The product of the hydrogenation reaction of following alkene with Pd catalyst should be determined:
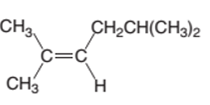
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form alkane. Certain metal catalyst Ni and Pd are used for this reaction.
(c)
Interpretation:
The product of the hydrogenation reaction of following alkene with Pd catalyst should be determined:
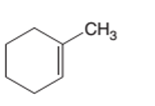
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form alkane. Certain metal catalyst Ni and Pd are used for this reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, & Biological Chemistry
- What alcohol is formed when each carbonyl compound is treated with H 2 and a Pd catalyst?arrow_forwardWhat alcohol is formed when the alkene CH3CH2CH=CH2 is treated with H2O in the presence of H2SO4?arrow_forwardDraw the products formed when ethylene oxide is treated with following reagent. [1] −OH; [2] H2Oarrow_forward
- Draw the products formed when ethylene oxide is treated with following reagent. [1] HC=C−; [2] H2Oarrow_forwardWhat alcohol is formed when each compound is treated with H 2 and a Pd catalyst?arrow_forwardDraw the products formed when ethylene oxide is treated with following reagent. [1] CH3S−; [2] H2Oarrow_forward
- What product is formed when p-dimethylbenzene (also called p-xylene) is treated with each reagent?arrow_forwardWhat alkane is formed when each alkene is treated with H2 and a Pd catalyst?arrow_forwardWhat alcohol is formed when the following compound is treated with H2 and a Pd catalyst?arrow_forward
- Draw the products formed when cis- and trans-but-2-ene are treated with CHCl3 and KOC(CH3)3arrow_forwardDraw the organic products formed when each alkyne is treated with two equivalents of HBr.arrow_forwardDraw the products formed when alkene reacts with these reagents: (i) H2 in the presence of Pd catalyst, (ii) HCl.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





