
Concept explainers
Interpretation: Starting material and reagents required to prepare the given epoxide need to be identified.
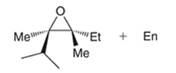
Concept introduction:
Preparation of epoxide-
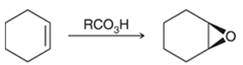
Peroxy acids generally used in this process are MCPBA and peroxyacetic acid. The formation of epoxide via peroxy acid is a stereospecific process; thus, cis substituents in alkene (starting material) remain at cis to each other in the epoxide (product). Similarly, trans substituents in alkene remain at trans to each other.
The preparation of epoxides from halohydrins can simply take place upon treatment with a strong base. Here, halohydrin can be formed from alkene after treatment with a halogen in the presence of water.
The reactions involved in the preparation are as follows
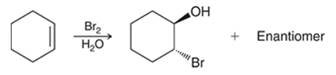
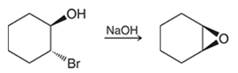
The above reaction takes place via intramolecular
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
ORGANIC CHEMISTRY-PRINT COMPANION (LL)
- Claisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forwardEthylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forwardProvide the missing reagents required for the following transformation: OH HO-arrow_forward
- Identify the best reagents to complete the following reaction. CI OHarrow_forwardIdentify A, B, and C, three intermediates in the synthesis of the pain reliever and anesthetic fentanyl.arrow_forwardShow how you would prepare the following from the indicated starting materials. More than one step is required. Show all the reaction products and the reaction conditions (reagents, temperatures, pressures). A from and from Br and Harrow_forward
- Provide the reagents necessary to carry out the following conversion. HO НОarrow_forwardWhat are the reagents of the following reaction? он ? ноarrow_forwardDetermining the Regioselectivity of Opening an Epoxide Ring What product is formed when 2,2-dimethyloxirane is treated with each set of reagents: −OCH3 followed by H2O, or CH3OH and H2SO4?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



