
Concept explainers
Draw a splitting diagram for
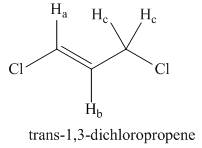
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
ORG.CHEMISTRY W/ACCESS+MODEL KIT PKG
Additional Science Textbook Solutions
Principles of General, Organic, Biological Chemistry
Fundamentals of Heat and Mass Transfer
Inorganic Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Organic Chemistry
Chemistry & Chemical Reactivity
- The R-branch of the 1Πu ← 1Σg+ transition of H2 shows a band head at the very low value of J = 1. The rotational constant of the ground state is 60.80 cm− 1. What is the rotational constant of the upper state? Has the bond length increased or decreased in the transition?arrow_forwardThe benzene radical anion has g = 2.0025. At what field should you search for resonance in a spectrometer operating at (i) 9.313 GHz, (ii) 33.80 GHz?arrow_forwardEstimate the spin-only molar susceptibility of CuSO4⋅5H2O at 25 °C.arrow_forward
- How many normal modes of vibration are there for (a)NO,. (b) N20. (c) cyclohexane. (d) hexane?arrow_forwardThe benzene radical anion has g = 2.0025. At whatfield should you search for resonance in a spectrometeroperating at (a) 9.302 GHz, (b) 33.67 GHz?arrow_forwardThe wavenumber of the J = 3 ← 2 rotational transition of 1H35Cl considered as a rigid rotor is 63.56 cm−1; what is the H–Cl bond length?arrow_forward
- The lowest energy transition (J=0 --> J=1) in teh pure rotational spectrum of 12C16O occures ar 3.8026 cm-1. Calculate the rotational constant, B in Joules (1cm-1 = 100hc = 1.987*10-23 J)arrow_forwardPredict the wavenumber of the 0 → 2 line of the rotational Raman spectrum of 14N2, for which B = 1.99 cm-1, when it is exposed to 336.732 nm laser radiation.arrow_forwardSuppose the C=O group in a peptide bond can be regarded as isolated from the rest of the molecule. Given the force constant of the bond in a carbonyl group is 908 N m-1, calculate the vibrational wavenumber of (a) 12C=16O.(b) 13C=16O.arrow_forward
- The molar absorption coefficient of a substance dissolved in hexane is known to be 227 dm3 mol−1 cm−1 at 290 nm. Calculate the percentage reduction in intensity when ultraviolet radiation of that wavelength passes through 2.00 mm of a solution of concentration 2.52 mmol dm−3.arrow_forwardCalculate the energy in KJ/mol associated with the 3.517-μm vibrational absorption bandof an aliphatic ketone.arrow_forwardEstimate the centrifugal distortion constant for 79Br81Br, for which ᷉ B = 0.0809 cm−1 and ᷉v = 323.2 cm−1. By what factor would the constant change when the 79Br is replaced by 81Br?arrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
