
Concept explainers
Interpretation:
A VB picture of the
Concept introduction:
Some chemical and structural features of molecules are unaccounted for by VB theory, most notably electron delocalization, or resonance, over three or more nuclei. The VB theory picture of a species that has resonance is inaccurate because it represents a single resonance structure, not the hybrid.
Answer to Problem 14.1P
The VB picture of the
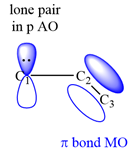
The structure of the resonance hybrid of allyl anion is
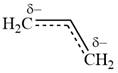
Explanation of Solution
All three carbon atoms in the allyl anion are
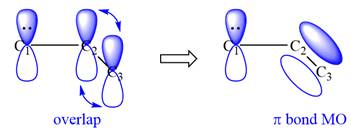
This corresponds to a double bond between C2 and C3, with a
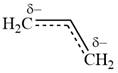
The VB theory picture of a given species represents a single resonance structure and not the hybrid.
Want to see more full solutions like this?
Chapter 14 Solutions
Get Ready for Organic Chemistry
- Diagram the pi molecular orbitals for the ion below. This drawing should be top view. + CH3 H ШІНarrow_forwardanswer the following problem and EXPLAIN YOUR ANSWER. ILLUSTRATE YOUR WORK ON A PAPER FOR BETTER UNDERSTANDINGarrow_forwardDetermine whether each structure is likely to be colored or not. If colored, indicating the extented conjugation by marking series of continuous sp2 hybridized atomsarrow_forward
- Orbital representation of the molecule in the image.arrow_forwardRe-sketch the diazene molecule and label all bond dipoles (with the arrow pointing from the most electronegative atom to the least electronegative atom). Show using vector sums that this geometric isomer of diazene is non-polar.arrow_forwardAccording to valence bond theory, what is the total number of sp2 hybridized orbitals on all the atoms in formaldehyde, CH2O? Sketch and explain why. According to valence bond theory, how many lone pairs of electrons reside in sp hybridized orbitals in formaldehyde? explain why?arrow_forward
- Draw the delocalized molecular orbitals for the following molecule. Are both bonds of the triple bond involved in the delocalized orbitals?arrow_forwardUsing cartoon representations, draw a molecular orbital mixing diagram for a CO bond. In your picture, consider the relative energies of C and O and how this changes the resulting bonding and antibonding molecular orbitals relative to a CC bond.arrow_forwardDetermine the correct hybridizaton (from left to right) about each interior atom in CH≡CCH2ClCH≡CCH2Cl.arrow_forward
- 2. In 2003 James Tour's research group at Rice University published a paper on the synthesis of the "nanokid" (shown below). [1] Draw in the hydrogens on each of the indicated atoms. [2] Identify the hybridization at each of the indicated atoms.arrow_forwardQuestion (i) is continuation to the previous questionarrow_forward7) How many sp hybridized carbon atom exist in the following structure? НО. NH2 04 01 O None O2 Озarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

