
Concept explainers
(a)
Interpretation:
The given molecule is to be identified as
Concept introduction:
Answer to Problem 14.11P
The given molecule is nonaromatic.
Explanation of Solution
The structure of the given molecule is
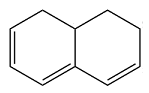
In the given compound, six carbon atoms are
Because of the
Therefore, this molecule is nonaromatic.
The presence of four
(b)
Interpretation:
The given molecule is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Huckel’s rule for aromaticity states that if a species is planar and possesses a
Answer to Problem 14.11P
The given molecule is aromatic.
Explanation of Solution
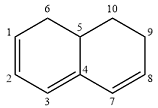
Structure of the given molecule is
In this molecule, six carbon atoms are
Therefore, the given molecule is aromatic.
The presence of six
(c)
Interpretation:
The given molecule is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Huckel’s rule for aromaticity states that if a species is planar and possesses a
Answer to Problem 14.11P
The given molecule is nonaromatic.
Explanation of Solution
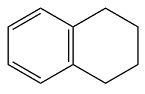
Structure of the given molecule is
In the given compound, four carbon atoms are
Therefore, the given molecule is nonaromatic.
The presence of one
(d)
Interpretation:
The given molecule is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Huckel’s rule for aromaticity states that if a species is planar and possesses a
Answer to Problem 14.11P
The given molecule is nonaromatic.
Explanation of Solution
Structure of the given molecule is
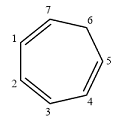
In the given compound, six carbon atoms are
Therefore, the given molecule is nonaromatic.
The presence of one
(e)
Interpretation:
The given molecule is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Huckel’s rule for aromaticity states that if a species is planar and possesses a
Answer to Problem 14.11P
The given molecule is nonaromatic.
Explanation of Solution
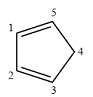
Structure of the given molecule is
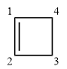
In the given compound, two carbon atoms are
Therefore, the given molecule is nonaromatic.
The presence of two
(f)
Interpretation:
The given molecule is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Huckel’s rule for aromaticity states that if a species is planar and possesses a
Answer to Problem 14.11P
The given molecule is nonaromatic.
Explanation of Solution
Structure of the given molecule is
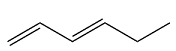
The molecule is not a cyclic system.
Therefore, the given molecule is nonaromatic.
Non-cyclic form of the given molecule indicates that the molecule is nonaromatic.
(g)
Interpretation:
The given molecule is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Huckel’s rule for aromaticity states that if a species is planar and possesses a
Answer to Problem 14.11P
The given molecule is aromatic.
Explanation of Solution
The structure of the given molecule is
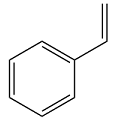
The molecule has a cyclic part with alternating single and double bonds. This means all ring atoms are
Therefore, this molecule is aromatic.
The presence of six
Want to see more full solutions like this?
Chapter 14 Solutions
Get Ready for Organic Chemistry
- are these aromatic, antiaromatic, or nonaromatic? Draw in all lonepairs and indicate the orbital each lone pair resides in.arrow_forwardWhich are aromatic species? Please circle correct species. (assume planarity unless otherwise noted)arrow_forwardBelow are two sets of resonance structures. Where applicable provide the missing curved arrow notation, lone pair electrons, and nonzero formal charge. Do not delete or add any bonds or atoms.arrow_forward
- Problem: (a) Draw six (6) constitutional isomers of C6H1202. (b) Which of the compounds you drew would be the most soluble in water? Explain your choice. (c) Which of the compounds you drew would be the most soluble in hexane? Explain your choice.arrow_forwardIn HF , neither H nor F holds a full formal charge of +1 or 1 . Organic chemists represent apartial charge using the Greek letter delta () . On the electron density map of the molecule HF above, add a + to one atom and a to the other to indicate which way the bond is polarized.arrow_forwardDraw Newman projections of the following molecules viewed from the direction of the blue arrows.arrow_forward
- Draw all reasonable resonance structures for the following cation. Thendraw the resonance hybrid.arrow_forwardidentify which is the most and least basic among the given. refer to the number and stability of their resonance structure.arrow_forwardDraw ALL REASONABLE resonance structures and the resonance hybrids of the compounds below.arrow_forward
- Draw the resonance structures for the following compound and circle the one that contributes most to the resonance hybrid. Why?arrow_forwardDraw the curved arrows to show the resonance structure for the following molecules. ( there are 3 molecules )arrow_forward3) Draw all resonance structures of the following compound. Order the structures by increasing stability.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
