
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
9th Edition
ISBN: 9781337580632
Author: Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 14.43E
Complete the following equations. If no reaction occurs, write “no reaction.”
a. 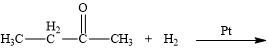
b. 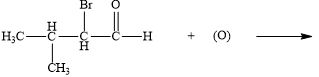
c. 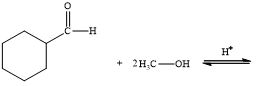
d. 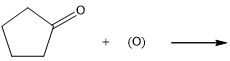
e. 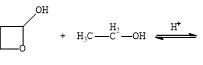
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
2. Differentiate acetic acid from hydrochloric acid in terms of.
a. Boiling point
b. Acidity
c. Solubility in ethyl alcohol
What is the product of the following reaction?
a.
b.
c.
d.
مر
Br
1)LDA, -780
2)EtBr
1.) H-B-
2.) H2O2, NaOH (xs)
a)
b.)
HO
d.)
Chapter 14 Solutions
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
Ch. 14 - Prob. 14.1ECh. 14 - Prob. 14.2ECh. 14 - Identify each of the following compounds as an...Ch. 14 - Identify each of the following compounds as an...Ch. 14 - Prob. 14.5ECh. 14 - Prob. 14.6ECh. 14 - Prob. 14.7ECh. 14 - Prob. 14.8ECh. 14 - Draw structural formulas and give IUPAC names for...Ch. 14 - Draw structural formulas and give IUPAC names for...
Ch. 14 - Each of the following names is wrong. Give the...Ch. 14 - Each of the following names is wrong. Give the...Ch. 14 - Prob. 14.13ECh. 14 - Prob. 14.14ECh. 14 - Prob. 14.15ECh. 14 - Explain why propane boils at 42C, whereas ethanal,...Ch. 14 - Use a dotted line to show hydrogen bonding between...Ch. 14 - Use a dotted line to show hydrogen bonding between...Ch. 14 - Prob. 14.19ECh. 14 - Prob. 14.20ECh. 14 - Prob. 14.21ECh. 14 - Prob. 14.22ECh. 14 - Prob. 14.23ECh. 14 - Prob. 14.24ECh. 14 - Label each of the following as acetals, ketals, or...Ch. 14 - Label each of the following as acetals, ketals, or...Ch. 14 - Label each of the following structures as a cyclic...Ch. 14 - Label each of the following structures as a...Ch. 14 - What two functional groups react to form the...Ch. 14 - Hemiacetals are sometimes referred to as potential...Ch. 14 - Complete the following statements: a. Oxidation of...Ch. 14 - Prob. 14.32ECh. 14 - Prob. 14.33ECh. 14 - Prob. 14.34ECh. 14 - Prob. 14.35ECh. 14 - Not all aldehyde give a positve Bendicts test....Ch. 14 - A stockroom assistant prepares three bottles, each...Ch. 14 - Glucose, the sugar present within the blood, gives...Ch. 14 - Fructose, present with glucose in honey, reacts...Ch. 14 - Prob. 14.40ECh. 14 - Prob. 14.41ECh. 14 - Complete the following equations. If no reaction...Ch. 14 - Complete the following equations. If no reaction...Ch. 14 - Describe the products that result when hydrogen...Ch. 14 - Prob. 14.45ECh. 14 - Draw structural formulas for the products of the...Ch. 14 - The following compounds are cyclic acetals or...Ch. 14 - The following compounds are cyclic acetals or...Ch. 14 - Write equations to show how the following...Ch. 14 - Prob. 14.50ECh. 14 - Identify the most important aldehyde and ketone...Ch. 14 - Using Table 14.3, name an aldehyde or ketone used...Ch. 14 - Prob. 14.53ECh. 14 - CH3COH(O)CH3COOHacetaldehydeaceticacid You need to...Ch. 14 - The addition of water to aldehydes and ketones...Ch. 14 - Prob. 14.56ECh. 14 - Formaldehyde levels above 0.10mg/1000L of ambient...Ch. 14 - In the IUPAC name for the following ketone, it is...Ch. 14 - Why can formaldehyde (CH2O) be prepared in the...Ch. 14 - Other addition reactions of aldehydes occur....Ch. 14 - Prob. 14.61ECh. 14 - Prob. 14.62ECh. 14 - Vanilla flavoring is either extracted from a...Ch. 14 - Prob. 14.64ECh. 14 - The use of acetone in laboratory experiments must...Ch. 14 - Prob. 14.66ECh. 14 - Prob. 14.67ECh. 14 - Which of the following would be classified as a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the complete Lewis electron dot structure for dichlorocarbene, :CCl₂. In the reaction of an alkene with dichlorocarbene, the dichlorocarbene is the: a. electrophile. b. Lewis base. The reaction of an alkene with dichlorocarbene is: a. regiospecific. b. Markovnikov. c. nucleophile. c. stereospecific. d. both b and c. d. non-Markovnikov.arrow_forwardWhat is the product of this reaction? Click on a letter A through D to answer. TSOH (cat) 8 888 А. В. C. D.arrow_forwardF. F C. 2. F F draw structurearrow_forward
- ORGANIC Chemistry Which are common names for carboxylic acid derivatives? A. Esters B. Ethers C. Formic Acid D. Acetic Anhydride Answers: A. C, D, and A B. D and B only C. C and D only D. None I think it is A, please answer and explain why. Thank youarrow_forwardwhich has the greatest acidity? a. 2-hexanol b. valeric acid c. naphtol d. ethyl propionate which produces effervescence when dissolved in 10% sodium bicarbonate? a. 2-hexanol b. valeric acid c. napththol d. ethyl propionatearrow_forwardDraw the following reactions and label it properly 1. stearic acid reaction with water chemical equation 2. Reaction with NaOH with benzoic acid 3. Reaction with NaOH with acetic acid 4. Reaction with NaOH with stearic acid 5. Reaction with sodium carbonate with acetic acidarrow_forward
- 1. Give at least four medicinal uses of salicylic acid. 2. What are the roles sulfuric acid in the synthesis of salicylic acid? 3. What are the effects of the carboxyl group to the activity of salicylic acid?arrow_forwardCH2 H3C H3C COOH Reagents e. PB13 f. NaCN then H3O* g. NBS, CCI4 h. CrO3, H2SO4 а. HBr b. Mg, ether c. CO2, ether then H3O* d. BH3, THF then H2O2, OH"arrow_forwardDoes alcohol, ketones, and aldehydes considered acidic or a basic?arrow_forward
- Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forwardC. Amides 1. Amidation a. Acetic acid + ammoniaarrow_forward2. Provide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY