
Concept explainers
Answer the following questions about each of the hydroxy
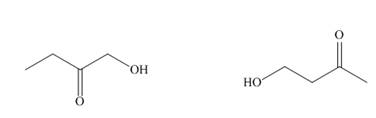
A B
a. What is the molecular ion in the mass spectrum?
b. What IR absorptions are present in the functional group region?
c. How many lines are observed in the
d. How many signals are observed in the
e. Give the splitting observed for each type of proton as well as its approximate chemical shift.
Trending nowThis is a popular solution!

Chapter 14 Solutions
ORGANIC CHEMISTRY (LOOSE)-MOLYMOD PKG.
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Living By Chemistry: First Edition Textbook
CHEMISTRY-TEXT
Organic Chemistry - Standalone book
- Consider isomeric alcohols A and B and mass spectra [1] and [2]. (a) Label the molecular ion and base peak in each spectrum. (b) Use thefragmentation patterns to determine which mass spectrum correspondsto isomer A and which corresponds to isomer B.arrow_forwardi need help in comparing ir spec of 1,4-dimethoxybenzene versues 1,4-dimethoxybenzene and what is present and absent in the the functional groups i made the ir specs belowarrow_forward(Eugenol) - Identify all the peaks from the IR spectrum. Be sure to list the cm-1 and the bond that corresponds to each peakarrow_forward
- What does the IR spectrum of 1,4-cyclohexanedione vs 1,2-cyclohexanedione look like?arrow_forward9) What would be the difference between the 4 alcohols in IR? They all show O-H, C-H sp3, and C-H aromatic bonds. The only way to tell which one is used would be by comparing the fingerprint regions to published IR spectra of each alcohol. Alcohol 2 has an aldehyde C-H group that could be seen between 2700-2800 and 2800-2900 cm-1 Only alcohol 3 will show O-H bond around 3300 cm-1 Only alcohol 1 will show C=C aromatic bonds around 1600 cm-1 10) Assuming 5.0 mL of alcohol 2 was used and 2.2 g of the product was isolated, what is the percent yield for this reaction? The product has a molar mass of 198.00 g/mol. 11) How could the percent yield be increased for this reaction? Add NaOH to neutralize the product as it forms Add a catalyst such as H2SO4 to help speed up the reaction Add a catalyst such as HCl to help speed up the reaction Cool the reaction on ice to increase the rate of reaction 12) Is there another way that the product of this reaction…arrow_forwardCircle the solvents from the following list that can be used with 1H NMR spectroscopy that do not interfere with the spectrum. carbon tetrachloride chloroform benzene-d6 hexachloroacetone acetonitrile acetone methylene chloride D2O DMF (dimethylformamide)arrow_forward
- Chemistry 1.Predict the IR signal in the 1-bromo-3-ethylbenzene. (Approximate wave number for each Functional groups) 2.Predict the 1HNMR signal in the 1-bromo-3-ethylbenzene. (No of signals, splitting pattern of signals, integration, etc)arrow_forwarda) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of spectra)? c) what is different about C=O and O-H bonds compared with C=C or C≡C bonds that show weaker signals?arrow_forwardThe IR and proton NMR of compound C12H18 shown below. A) what is it U value? B) How many equivalent H’s per molecule are responsible for singlet peak at 1.35 ppm in the proton NMR C) How many equivalent H’s per molecule are responsible for singlet peak at 2.35 ppm D) Draw the structure base on the Ir and NMrarrow_forward
- 4. Match the correct compound to the given IR spectra. On the IR spectrum, label the absorptions with appropriate functional groups.arrow_forwardLabel all relevant peaks that are above the fingerprint region (>1500 cm-1) in all the IR spectra given below with the functional group that those peaks are representative of (make sure not to forget the aldehyde C–H bonds, which many IR charts do not list). Based on the table you completed with predicted Diagnostic IR peaks for each compound, and based on the IR spectra you had labeled, match each compound with its spectrum.arrow_forwardDirectly label the bonds/functional groups on the IR spectrum. Label all major peaksarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





