
CHEMISTRY 1407 >CUSTOM PACKAGE<
18th Edition
ISBN: 9781323745632
Author: Central Texas
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 14.61APP
Draw the condensed structural or line-angle formulas for the products of
the following: (14.2, 14.3, 14.6)
a.
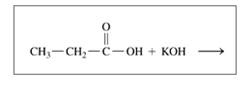
b.
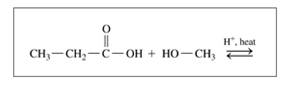
c.
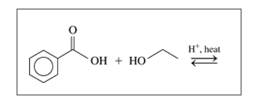
d.
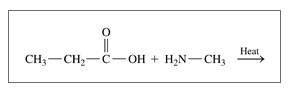
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following statements is true16. lodoform, which has a molecular formula of CHal, is indicative that a ketomethyl group is present. 17. Based on the physical property, hydroquinone has a lower boiling point compared to catechol. 18. When FeCl3 is used in visualizing aspirin in thin layer chromatography, the resulting spot is red in color.
a.Identify the functional groups in the ball-and-stick model of neral, a compound with a lemony odor isolated from lemongrass.
b. Draw a skeletal structure of a constitutional isomer of neral that should be more water soluble.
c.Label the most electrophillic carbon atom.
Esterification Reaction:
12.00 grams of acetic acid is reacted with methanol and sulfuric acid catalyst under heated conditions. What amount (in grams) of the methanol is required for the reaction and what is the theoretical yield of the methyl acetate product? Include a balanced equation.
Chapter 14 Solutions
CHEMISTRY 1407 >CUSTOM PACKAGE<
Ch. 14.1 - What carboxylic acid is responsible for the pain...Ch. 14.1 - What carboxylic acid is found in vinegar?Ch. 14.1 - Prob. 14.3PPCh. 14.1 - Prob. 14.4PPCh. 14.1 - Draw the condensed structural formulas for a and b...Ch. 14.1 - Draw the condensed structural formulas for a and b...Ch. 14.2 - Identify the compound in each group that is most...Ch. 14.2 - Prob. 14.8PPCh. 14.2 - Prob. 14.9PPCh. 14.2 - Prob. 14.10PP
Ch. 14.2 - Prob. 14.11PPCh. 14.2 - Prob. 14.12PPCh. 14.2 - Prob. 14.13PPCh. 14.2 - Prob. 14.14PPCh. 14.3 - Prob. 14.15PPCh. 14.3 - Prob. 14.16PPCh. 14.3 - Prob. 14.17PPCh. 14.3 - Prob. 14.18PPCh. 14.3 - Prob. 14.19PPCh. 14.3 - Prob. 14.20PPCh. 14.3 - Prob. 14.21PPCh. 14.3 - Prob. 14.22PPCh. 14.3 - Prob. 14.23PPCh. 14.3 - Prob. 14.24PPCh. 14.4 - What are the products of the acid hydrolysis of an...Ch. 14.4 - Prob. 14.26PPCh. 14.4 - Prob. 14.27PPCh. 14.4 - Prob. 14.28PPCh. 14.5 - Prob. 14.29PPCh. 14.5 - Prob. 14.30PPCh. 14.5 - Prob. 14.31PPCh. 14.5 - Prob. 14.32PPCh. 14.5 - Prob. 14.33PPCh. 14.5 - Prob. 14.34PPCh. 14.5 - Prob. 14.35PPCh. 14.5 - Prob. 14.36PPCh. 14.5 - Prob. 14.37PPCh. 14.5 - Prob. 14.38PPCh. 14.6 - Prob. 14.39PPCh. 14.6 - Prob. 14.40PPCh. 14.6 - Prob. 14.41PPCh. 14.6 - Prob. 14.42PPCh. 14.6 - Prob. 14.43PPCh. 14.6 - Prob. 14.44PPCh. 14.6 - Draw the condensed structural or line-angle...Ch. 14.6 - Draw the condensed structural or line-angle...Ch. 14.6 - a. Identify the functional groups in dicyclanil....Ch. 14.6 - a. Identify the functional groups in enrofloxacin....Ch. 14 - Prob. 14.49UTCCh. 14 - Prob. 14.50UTCCh. 14 - The ester methyl butanoate has the odor and flavor...Ch. 14 - Prob. 14.52UTCCh. 14 - Phenylephrine is the active ingredient in some...Ch. 14 - Melatonin is a naturally occurring compound in...Ch. 14 - Prob. 14.55UTCCh. 14 - Prob. 14.56UTCCh. 14 - Prob. 14.57APPCh. 14 - 14.58 Write the IUPAC and common names, if any,...Ch. 14 - Prob. 14.59APPCh. 14 - Prob. 14.60APPCh. 14 - Draw the condensed structural or line-angle...Ch. 14 - Prob. 14.62APPCh. 14 - Prob. 14.63APPCh. 14 - 14.64 Draw the condensed structural or line-angle...Ch. 14 - Prob. 14.65APPCh. 14 - 14.66 Write the common name and classify each of...Ch. 14 - Prob. 14.67APPCh. 14 - Draw the condensed structural or line-angle...Ch. 14 - Prob. 14.69APPCh. 14 - Prob. 14.70APPCh. 14 - Write the IUPAC name for each of the following:...Ch. 14 - Prob. 14.72APPCh. 14 - Prob. 14.73APPCh. 14 - Prob. 14.74APPCh. 14 - Prob. 14.75APPCh. 14 - Toradol is used in dentistry to relieve pain....Ch. 14 - Prob. 14.77CPCh. 14 - Draw the line-angle formula and write the IUPAC...Ch. 14 - Prob. 14.79CPCh. 14 - Prob. 14.80CPCh. 14 - Prob. 14.81CPCh. 14 - Prob. 14.82CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Review the opening photograph about chocolate (which shows the structure of an active ingredient, theobromine) and then answer the following questions. (a) How do theobromine and caffeine differ structurally? (b) A 5.00-g sample of Hersheys cocoa contains 2.16% theobromine. What is the mass of the compound in the sample?arrow_forwardBACTROBAN ointment contains 2% w/w mupirocin. H ow many grams of a polyethylene glycol ointment base must be mixed with the contents of a 22-g tube of the BACT ROBAN ointment to prepare one having a concentration of 5 mg/g?arrow_forwardLinolenic acid (Table 10.2) and stearidonic acid are omega-3 fatty acids, unsaturated fatty acids that contain the first double bond located at C3, when numbering begins at the methyl end of the chain. Predict how the melting point of stearidonic acid compares with the melting points of linolenic and stearic acids. A current avenue of research is examining the use of soybean oil enriched in stearidonic acid as a healthier alternative to vegetable oils that contain fewer degrees of unsaturation.arrow_forward
- 1.T/F California wines, unlike those in France, are not allowed to be blends of more than two varieties of grape. Select one: True Falsearrow_forwardThe three-carbon diol used in antifreeze is It is nontoxic and is used as a moisturizing agent in foods. Oxidation of this substance within the liver produces pyruvic acid, which can be used by the body to supply energy. Give the structure of pyruvic acid.arrow_forward5.00 grams of benzoic acid is reacted with ethanol and sulfuric acid catalyst under heated conditions. What amount (in grams) of the ethanol is required for the reaction and what is the theoretical yield of the ethyl benzoate product? Include a balanced equation.arrow_forward
- The melting point of unsaturated carboxylic acid increase with increase of duble bonds. Select the correct answer: a. True b. Falesarrow_forwardWhat is the preferred IUPAC name of the compound in Figure 12. [Numbers are separated by commas and numbers and letters by hyphens. Use lowercase letters and do not use spaces.] * Your answerarrow_forwardEq 12arrow_forward
- Cyclohexene has a boiling point higher than water. Group of answer choices True Falsearrow_forwardDigitalis is a preparation made from the dried seeds and leaves of the purple foxglove, Digitalis purpurea, a plant native to southern and central Europe and cultivated in the United States. The preparation is a mixture of several active components, including digitalin. Digitalis is used in medicine to increase the force of myocardial contraction and as a conduction depressant to decrease heart rate (the heart pumps more forcefully but less often).arrow_forwardThe amount of tartaric acid is responsible for the tartness of wine and controls the acidity of the wine. Tartaric acid also plays a very significant role in the overall taste, feel and color of a wine. Tartaric acid is a diprotic organic acid The chemical formula for tartaric acid is C4H6O6 and its structural formula is HO2CCH(OH)CH(OH)CO2H. A 50.00 mL sample of a white dinner wine required 21.48 mL of 0.03776 M NaOH to achieve a faint pink color. Express the acidity of the wine in terms of grams of tartaric acid, H2C4H4O6 (M. M. = 150.10) per 100 mL of wine. Assume that the two acidic hydrogens are titrated at the end point. MM H2C4H4O6 = 150.10 MM NaOH = 40.00 Below is the balanced chemical equation for this titration.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY