
Concept explainers
Propose a structure consistent with each set of data.
a.
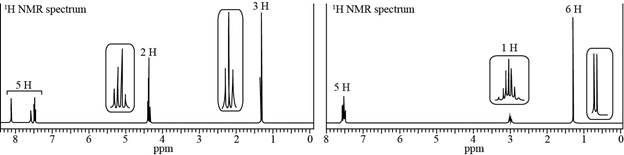
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry For Changing Times (14th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Chemistry: A Molecular Approach
Introductory Chemistry (6th Edition)
- Figures 2 to 4 show the IR, 1H NMR and 13C NMR spectra of a compound with formula C3H5N 2a) Identify bands A and B in the IR spectrumarrow_forwardThe molar absorptivity of 2,5-dimethyl-2,4-hexadiene in methanol is 13,100 M-1cm-1. What concentration of this diene in methanol is required to give an absorbance of 1.6? Assume a light path of 1.00 cm. Calculate concentration in these units. Q.)Milligrams per milliliterarrow_forwardDraw the structure of the compound that produced the spectra below. The infrared spectrum has strong bands at 1720 and 1738 cm-1.arrow_forward
- Propose structures for compounds that fit the following data: (a) A ketone with M+=86 and fragments at m/z=71 and m/z=43 (b) An alcohol with M+=88 and fragments at m/z=73, m/z=70, and m/z=59arrow_forwardA compound shows strong, very broad IR absorption in the region 3300–3600 cm-1 and strong, sharp absorption at 1715 cm-1. What functional group accounts for both of these absorptions?arrow_forwardPropose a structure consistent with each set of data. C8H10 : IR absorptions at 3108–2875, 1606, and 1496 cm−1arrow_forward
- Explain the order of chemical shifts of the carbon atoms in the 13C spectra of cyclohexanone (fig 22.6 ) and adipic acid (fig .22.9) attached below .arrow_forwardThe molar absorptivity of 2,5-dimethyl-2,4-hexadiene in methanol is 13,100 M-1cm-1. What concentration of this diene in methanol is required to give an absorbance of 1.6? Assume a light path of 1.00 cm. Calculate concentration in these units. Q.) Moles per literarrow_forward1. True or False a. UV-Vis spectroscopy normally reports data in the from of bands rather than single peaks because of overlapping electronic transitions that are being recorded by the detector. (T/F) b. When using linear regression to translate absorption data using Beer's Law, the y-intercept (+b) of the linear equation represents the path length. (T/F) c. Phosphorescent materials give a glowing effect because the electrons remain at an excited state for much longer, which is the cause for the "glow". (T/F)arrow_forward
- Identify the structure and explain the reasoning to why the strucutre matches a doublet at 9.7 ppm and a multiplet around 3.8 ppm. The IR spectrum shows a strong band around 3400 cm-1 and another at 1730 cm-1.arrow_forward2 The IR and 1H-NMR spectra of a compound with molecular formula C4H7ClO2 are shown below. Your objective as a group is to propose a structure for this compound, explaining how you reach your decision. Using all the information you have been given, in a post with others in your group share your initial ideas about the possible structure of the compound. Then use comments to interact with the other students in the group and propose a final answer to the problem. In the comment phase, you should comment on the postings of at least two other students.arrow_forwardPart 3A Set 1. Can IR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. What significant absorptions would be observed in the IR spectrum?arrow_forward

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

