
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 14.83SP
Consider the following concentration-time data for the decomposition reaction
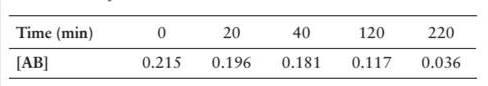
- Determine the order of the reaction and the value of therate constant.
- What is the molarity of AB after a reaction time of192 min?
- What is the time (in minutes) when the AB concentration reaches a value of 0.0250 M?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the reaction: H2 (g) +I2 (g) → 2 HI (g)
A chemist performed an experiment and monitored the concentration of I2 during the course of the reaction. The red line in the
graph below represents the results obtained.
Which line in the plot would best represent how the concentration of HI changes during the course of the reaction?
Time (s)
Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer.
a
a
b
b
d
d
е е
Concentration (M)
2C1₂0, (g) → 2Cl₂ (g) +50₂ (g)
He fills a reaction vessel with C1₂O5 and measures its concentration as the reaction proceeds:
time
(seconds)
0
0.10
0.20
0.30
[C₁₂05]
0.0500M
0.0322M
0.0207 M
0.0133M
0.40 0.00858M
Use this data to answer the following questions.
Write the rate law for this reaction.
Calculate the value of the rate constant k.
Round your answer to 2 significant digits. Also be
sure your answer has the correct unit symbol.
rate = k [C₁₂0₂]
k =
0
x10
□・□
X
Consider the reaction data.
A⟶products
? (?)
? (?−1)
325
0.391
575
0.735
What two points should be plotted to graphically determine the activation energy of this reaction? To avoid rounding errors, use at least three significant figures in all values.
x1=
y1=
x1=
x2=
Determine the rise, run, and slope of the line formed by these points.
What is the activation energy of this reaction?
Chapter 14 Solutions
CHEMISTRY-TEXT
Ch. 14 - Prob. 14.1PCh. 14 - Prob. 14.2ACh. 14 - The rate law for the reaction...Ch. 14 - Prob. 14.4ACh. 14 - The initial rates listed in the following...Ch. 14 - Prob. 14.6ACh. 14 - Prob. 14.7PCh. 14 - Prob. 14.8ACh. 14 - Prob. 14.9PCh. 14 - Prob. 14.10A
Ch. 14 - Prob. 14.11PCh. 14 - Prob. 14.12ACh. 14 - Prob. 14.13PCh. 14 - Prob. 14.14ACh. 14 - Consider the first-order decomposition of H2O2...Ch. 14 - Prob. 14.16ACh. 14 - Hydrogen iodide gas decomposes at 410 °C:...Ch. 14 - Prob. 14.18ACh. 14 - Thereaction NO2(g)+CO(g)NO(g)+CO2(g) occurs in one...Ch. 14 - Prob. 14.20ACh. 14 - Prob. 14.21PCh. 14 - Apply 13.22 The rate of the reaction...Ch. 14 - Prob. 14.23PCh. 14 - Prob. 14.24ACh. 14 - Prob. 14.25PCh. 14 - Prob. 14.26ACh. 14 - Prob. 14.27PCh. 14 - Prob. 14.28ACh. 14 - The following mechanism has been proposed for the...Ch. 14 - Prob. 14.30ACh. 14 - Prob. 14.31PCh. 14 - Draw a potential energy diagram for the mechanism...Ch. 14 - Prob. 14.33PCh. 14 - Given the mechanism for an enzyme-catalyzed...Ch. 14 - Prob. 14.35PCh. 14 - Prob. 14.36PCh. 14 - At high substrate concentrations, the rate...Ch. 14 - Chymotrypsin is a digestive enzyme component of...Ch. 14 - Prob. 14.39CPCh. 14 - Prob. 14.40CPCh. 14 - Prob. 14.41CPCh. 14 - Prob. 14.42CPCh. 14 - Prob. 14.43CPCh. 14 - Prob. 14.44CPCh. 14 - Prob. 14.45CPCh. 14 - Prob. 14.46CPCh. 14 - Prob. 14.47CPCh. 14 - Prob. 14.48CPCh. 14 - Prob. 14.49CPCh. 14 - Use the data in Table 13.1 to calculate the...Ch. 14 - 13.50 Use the data in Table 13.1 to calculate the...Ch. 14 - Prob. 14.52SPCh. 14 - Prob. 14.53SPCh. 14 - From the plot of concentrationtime data in Figure...Ch. 14 - Prob. 14.55SPCh. 14 - Prob. 14.56SPCh. 14 - Prob. 14.57SPCh. 14 - Prob. 14.58SPCh. 14 - Prob. 14.59SPCh. 14 - Prob. 14.60SPCh. 14 - Prob. 14.61SPCh. 14 - Prob. 14.62SPCh. 14 - Prob. 14.63SPCh. 14 - Prob. 14.64SPCh. 14 - Prob. 14.65SPCh. 14 - Prob. 14.66SPCh. 14 - Prob. 14.67SPCh. 14 - The oxidation of iodide ion by hydrogen peroxide...Ch. 14 - Prob. 14.69SPCh. 14 - At 500 °C, cyclopropane (C3H6) rearranges to...Ch. 14 - The rearrangement of methyl isonitrile (CH3NC) to...Ch. 14 - What is the half-life (in minutes) of the reaction...Ch. 14 - Prob. 14.73SPCh. 14 - Prob. 14.74SPCh. 14 - Hydrogen iodide decomposes slowly to H2 and I2 at...Ch. 14 - What is the half-life (in minutes) of the reaction...Ch. 14 - Prob. 14.77SPCh. 14 - At 25 °C, the half-life of a certain first-order...Ch. 14 - The decomposition of N2O5 is a first-order...Ch. 14 - Prob. 14.80SPCh. 14 - Prob. 14.81SPCh. 14 - Prob. 14.82SPCh. 14 - Consider the following concentration-time data for...Ch. 14 - Trans-cycloheptene (C7H12), a strained cyclic...Ch. 14 - Thelight-stimulatedconversionof 11-cis-retinalto...Ch. 14 - Why don't all collisions between reactant...Ch. 14 - Prob. 14.87SPCh. 14 - Prob. 14.88SPCh. 14 - Prob. 14.89SPCh. 14 - The values of Ea=183 kJ/mol and E=9 kJ/mol have...Ch. 14 - Prob. 14.91SPCh. 14 - Consider three reactions with different values of...Ch. 14 - Prob. 14.93SPCh. 14 - Rate constants for the reaction...Ch. 14 - Prob. 14.95SPCh. 14 - Prob. 14.96SPCh. 14 - Prob. 14.97SPCh. 14 - If the rate of a reaction increases by a factor of...Ch. 14 - Prob. 14.99SPCh. 14 - Prob. 14.100SPCh. 14 - Rate constants for the reaction...Ch. 14 - Prob. 14.102SPCh. 14 - Poly(ethylene terephthalate) is a synthetic...Ch. 14 - Prob. 14.104SPCh. 14 - Prob. 14.105SPCh. 14 - Prob. 14.106SPCh. 14 - The following mechanism has been proposed for the...Ch. 14 - Prob. 14.108SPCh. 14 - Prob. 14.109SPCh. 14 - The thermal decomposition of nitryl chloride,...Ch. 14 - The substitution reactions of molybdenum...Ch. 14 - The reaction 2NO2(g)+F2(g)2NO2F(g) has a second...Ch. 14 - The decomposition of ozone in the upper atmosphere...Ch. 14 - Prob. 14.114SPCh. 14 - The following mechanism has been proposed for the...Ch. 14 - Prob. 14.116SPCh. 14 - Prob. 14.117SPCh. 14 - Prob. 14.118SPCh. 14 - Prob. 14.119SPCh. 14 - Prob. 14.120SPCh. 14 - Prob. 14.121SPCh. 14 - Prob. 14.122SPCh. 14 - Prob. 14.123SPCh. 14 - Consider the reaction 2NO(g)+O2(g)2NO2(g) . The...Ch. 14 - Concentration-time data for the conversion of A...Ch. 14 - Prob. 14.126MPCh. 14 - Prob. 14.127MPCh. 14 - Prob. 14.128MPCh. 14 - Prob. 14.129MPCh. 14 - Prob. 14.130MPCh. 14 - Prob. 14.131MPCh. 14 - Prob. 14.132MPCh. 14 - Prob. 14.133MPCh. 14 - Prob. 14.134MPCh. 14 - Polytetrafluoroethylene (Teflon) decomposes when...Ch. 14 - The reaction A is first order in the reactant A...Ch. 14 - Prob. 14.137MPCh. 14 - A 1.50 L sample of gaseous HI having a density of...Ch. 14 - The rate constant for the decomposition of gaseous...Ch. 14 - The rate constant for the first-order...Ch. 14 - Prob. 14.141MPCh. 14 - Prob. 14.142MPCh. 14 - At 791 K and relatively low pressures, the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The label on a bottle of 3% (by volume) hydrogen peroxide, H2O2, purchased at a grocery store, states that the solution should be stored in a cool, dark place. H2O2decomposes slowly over time, and the rate of decomposition increases with an increase in temperature and in the presence of light. However, the rate of decomposition increases dramatically if a small amount of powdered MnO- is added to the solution. The decomposition products are H2O and O2. MnO2 is not consumed in the reaction. Write the equation for the decomposition of H2O2. What role does MnO2 play? In the chemistry lab, a student substituted a chunk of MnO2 for the powdered compound. The reaction rate was not appreciably increased. WTiat is one possible explanation for this observation? Is MnO2 part of the stoichiometry of the decomposition of H2O2?arrow_forwardHow do chemists envision reactions taking place in terms of the collision model for reactions? Give an example of a simple reaction and how you might envision the reaction’s taking place by means of a collision between the molecules.arrow_forwardWhich reaction mechanism assumptions are unimportant in describing simple ionic reactions between cations and anions? Why?arrow_forward
- Substances that poison a catalyst pose a major concern for many engineering designs, including those for catalytic converters. One design option is to add materials that react with potential poisons before they reach the catalyst. Among the commonly encountered catalyst poisons are silicon and phosphorus, which typically form phosphate or silicate ions in the oxidizing environment of an engine. Group 2 elements are added to the catalyst to react with these contaminants before they reach the working portion of the catalytic converter. If estimates show that a catalytic converter will be exposed to 625 g of silicon during its lifetime, what mass of beryllium would need to be included in the design?arrow_forwardThe element Co exists in two oxidation states, Co(II) and Co(III), and the ions form many complexes. The rate at which one of the complexes of Co(III) was reduced by Fe(II) in water was measured. Determine the activation energy of the reaction from the following data: T(K) k (s-1) 293 0.054 298 0.100arrow_forwardDetermine rxnH 25 C for the following reaction: NO g O2 g NO2 g This reaction is a major participant in the formation of smog.arrow_forward
- For the reaction of crystal violet with NaOH(aq), the measured rate of reaction is 1.27 106 mol L1 s1 when the concentration of crystal violet cation is 4.13 105 mol/L. (a) Estimate how long it will take for the concentration of crystal violet to drop from 4.30 105 mol/L to 3.96 105 mol/L. (b) Could you use the same method to make an accurate estimate of how long it would take for the concentration of crystal violet to drop from 4.30 105 mol/L to 0.43 105 mol/L? Explain why or why not.arrow_forward. How does the collision model account for the fact that a react ion proceeds faster when the concentrations of the reactants are increased?arrow_forwardFor the following reaction profile, indicate a. the positions of reactants and products. b. the activation energy. c. E for the reaction.arrow_forward
- You carry out the following reaction by introducing N2O4 into an evacuated flask and observing the concentration change of the product over time. N2O4(g)2NO2(g) Which one of the curves shown here reflects the data collected for this reaction?arrow_forwardAccount for the relationship between the rate of a reaction and its activation energy.arrow_forwardThe rate constant, k, at 25 C is 0.27/h for the reaction Pt(NH3)2Cl2(aq) + H2O() [Pt(NH3)2(H2O)Cl]+(aq) + Cl(aq) and the rate equation is Reaction rate = k[Pt(NH3)2C12] Calculate the rate of reaction when the concentration of Pt(NH3)2Cl2 is 0.020 M.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY