
Interpretation:
The magnitude of the electric dipole moment of methanal has to be calculated.
Concept introduction:
The quantity that is used to measure the uneven distribution of electrons around the atoms of a compound that possess the covalent bonds is known as dipole moment. The electric dipole moment is basically a measurement of the separation of negative and positive electrical charges. It is denoted by
Answer to Problem 14A.1ST
The magnitude of the electric dipole moment of methanal is
Explanation of Solution
The given illustration of methanal or formaldehyde is shown below.
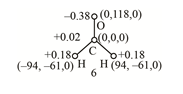
Figure 1
The positions of the given atoms are represented by
The value of charge is
The equation that s used to calculate the electric dipole is given below.
The value of
The value of
Substitute
The structure of methanol is planar, so
Substitute
Therefore, the magnitude of the electric dipole moment of methanal is
Want to see more full solutions like this?
Chapter 14 Solutions
PHYSICAL CHEMISTRY-WEBASSIGN
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





