
Organic Chemistry, 12e Binder Ready Version Study Guide / Student Solutions Manual
12th Edition
ISBN: 9781119077336
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 30P
For each of the pairs below, predict specific aspects in their
(a)
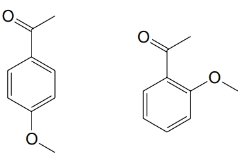
(b)
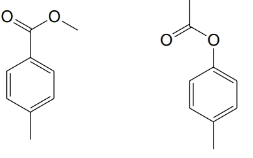
(c)
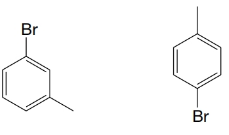
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compound b is a derivative of compound a. Comparing ¹H NMR of a and b, we found that two signals shown in
compound a move to downfield around 7.38 ppm and 8.42 ppm in compound b. Identify the two protons that most likely
give rise to these signals and justify your decision. (3 pts)
(a)
Ĵ
(b)
1. (a) How many 'H NMR signals will the following compound give? (b) What are the
relative intensities for those signals? List them in an order of increasing values.
Br-
Br
How could 1H NMR spectroscopy be used to distinguish among isomers A, B, and C?
Chapter 14 Solutions
Organic Chemistry, 12e Binder Ready Version Study Guide / Student Solutions Manual
Ch. 14 - PRACTICE PROBLEM 14.1 Provide a name for each of...Ch. 14 - Prob. 2PPCh. 14 - Prob. 3PPCh. 14 - Practice Problem 14.4 Apply the polygon-and-circle...Ch. 14 - Practice Problem 14.5 Apply the polygon-and-circle...Ch. 14 - Practice Problem 14.6 1,3,5-Cycloheptatriene is...Ch. 14 - Prob. 7PPCh. 14 - Prob. 8PPCh. 14 - Practice Problem 14.9 In 1967 R. Breslow (of...Ch. 14 - Prob. 10PP
Ch. 14 - Practice Problem 14.11 In addition to a signal...Ch. 14 - PRACTICE PROBLEM 14.12
Azulene has an appreciable...Ch. 14 - Practice Problem 14.13 (a) The -Sh group is...Ch. 14 - Practice Problem 14.14
Explain how NMR...Ch. 14 - PRACTICE PROBLEM 14.15 Four benzenoid compounds,...Ch. 14 - Prob. 16PCh. 14 - Write structural formulas and give acceptable...Ch. 14 - Prob. 18PCh. 14 - Prob. 19PCh. 14 - Prob. 20PCh. 14 - Which of the hydrogen atoms shown below is more...Ch. 14 - 14.22 The rings below are joined by a double bond...Ch. 14 - Prob. 23PCh. 14 - 14.24 (a) In 1960 T. Katz (Columbia University)...Ch. 14 - Prob. 25PCh. 14 - Prob. 26PCh. 14 - 14.27 5-Chloro-1,3-cyclopentadiene (below)...Ch. 14 - Prob. 28PCh. 14 - Furan possesses less aromatic character than...Ch. 14 - 14.30 For each of the pairs below, predict...Ch. 14 - Assign structures to each of the compounds A, B,...Ch. 14 - Prob. 32PCh. 14 - Give a structure for compound F that is consistent...Ch. 14 - Prob. 34PCh. 14 - Prob. 35PCh. 14 - The IR and 1H NMR spectra for compound X(C8H10)...Ch. 14 - Prob. 37PCh. 14 - Prob. 38PCh. 14 - 14.39 Given the following information, predict the...Ch. 14 - Consider these reactions: The intermediate A is a...Ch. 14 - Prob. 41PCh. 14 - Compound E has the spectral features given below....Ch. 14 - Draw all of the molecular orbitals for...Ch. 14 - Prob. 1LGPCh. 14 - Prob. 2LGPCh. 14 - 3. The NMR signals for the aromatic hydrogens of...Ch. 14 - Prob. 4LGPCh. 14 - Prob. 5LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
What dipeptides would be formed by heating a mixture of valine and N-protected leucine?
Organic Chemistry (8th Edition)
1.3 Obtain a bottle of multivitamins and read the list of ingredients. What are four chemicals from the list?
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
9.1 Calculate the total mass of the reactants and the products for each of the following equations:
Basic Chemistry (5th Edition)
3.1 The reaction between reactant A (blue spheres) and reactant B (red spheres) is shown in the
following diag...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (a) The 'H-NMR spectrum of cyclobutanone shows two signals - signal A at 3.00 ppm and signal B at 1.95 ppm. Give the multiplicity of each signal. cyclobutanone (b) When cyclobutanone is treated with D20 and NaOD, the only signal observable in the 1H-NMR is a singlet at 2.00 ppm. Explain why this is the case. [Note: Deuterium atoms do not display signals in the TH-NMR spectrum]arrow_forward2. Each compound below has only 1 peak in its ¹H NMR spectrum. Use the NMR tables to predict the chemical shift range (ppm) where each compound should absorb. (d) (a) (b) (c) CH₂Cl2 (e) zl H H (f) CH3OCH 3arrow_forwardDraw a structure for the compound, C3H5Br, that fits the following 'H NMR data: d 2.32 (3H, singlet) ô 5.35 (1H, broad singlet) ô 5.54 (1H, broad singlet) You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. In cases where there is more than one answer, just draw one. C opy aste C ChemDoodlearrow_forward
- Treatment of compound C (molecular formula C4H8O) with C6H5MgBr, followed by H2O, affords compound D (molecular formula C10H14O). Compound D has a strong peak in its IR spectrum at 3600–3200 cm−1. The 1H NMR spectral data of C and D are given. What are the structures of C and D? Compound C signals at 1.3 (singlet, 6 H) and 2.4 (singlet, 2 H) ppm Compound D signals at 1.2 (singlet, 6 H), 1.6 (singlet, 1 H), 2.7 (singlet, 2 H), and 7.2 (multiplet, 5 H) ppmarrow_forward3. How would you distinguish between each of the following pair of compounds by proton nmr? a) b)arrow_forward2 (a) In the following reaction, PCC A (i) Draw the structure of compound A. (ii) Explain using the IR spectra to confirm that the reaction is completed. (iii) Identify the molecular ion peak for compound A. (iv) Fragmentation of A shows a peak at m/z 111. Draw the possible cation for this peak. (v) Determine the resonance structure of this cation.arrow_forward
- Identify the lettered compounds in the following reaction scheme. Compounds F, G, and K are isomers of molecular formula C13H18O. How could 1H NMR spectroscopy distinguish these three compounds from each other?arrow_forwardThe 1H and 13C NMR spectra of compound A, C8H9Br are shown below. Answer the following questions. 1(a) Degree of the unsaturation of this compound is = , 1(b) The derived unsaturation number indicates that compound has ............= 1(c) Two peaks in between 6.5 - 8.0 δ indicate that compound is= 1(d) According to the splitting pattern of the peak at 1.20 δ and 2.58 δ indicates that compound has a .................. group= 1(e) According to the 1H NMR spectrum the number of nonequivalent aromatic proton sets in the compound = 1(f) According to the 13C NMR, the number of nonequivalent carbons in the compound is = 1(g) According to your answer in Q 1(f) the compound has a plane of symmetry Yes or NO = 1(h) The IUAC name for this unknown compound isNOT TOO SURE ABOUT MY ANSWERS, PLEASE CORRECT ME IF I'M WRONGarrow_forward1. Show how you would distinguish among the following three isomers (7 pts) vol or ol H OCH3 OH H3CO HO H3C (a) (b) (c) a) Using ¹H NMR spectroscopy and no other information. (4 pts) b) Using ¹³C NMR, including DEPT, and no other information.(3 pts)arrow_forward
- A different unknown has an FTIR with a very intense peak at 1735 cm. Its 13C NMR spectrum exhibits a peak at 169.3 ppm, and the 'H NMR spectrum shows a quartet at 4.07 ppm. Which of the following compounds is the unknown most likely to be? (a) (b) (c) (d) (e) O compound (a) O compound (d) compound (e) compound (c) compound (b) (0)arrow_forwardYou are given an unknown compound B with the molecular formula C5H12O and you take an 1H NMR of B (See below). You were told that B formed when A was reacted with a catalytic amount of H2SO4 and an excess of H2O, A formed. An IR spectra of A is below. Identify A and B in the spaces belowarrow_forwardCompound A has molecular formula C5H8Br4 but shows only one singlet in the 1H-NMR spectrum. Suggest a structure for A and explain your reasoning.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY