
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 40P
Consider these reactions:
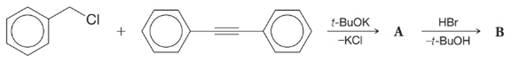
The intermediate A is a covalently bonded compound that has typical
What are the structures of A and B?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compound A is treated with a mixture of nitric and sulfuric acids to generate Compound B. The 1H-NMR spectrum of B shows two singlets, one at 2.52 pm and one at 8.13 ppm. The 13C-NMR spectrum of B shows five signals. The mass spectrum of B shows a peak at m/z = 260 and another peak at m/z = 262; the relative height of the two peaks is 1:1 respectively.
- Identify compound B, explaining your reasoning
19. A compound with molecular formula C6H12O2 exhibits two singlets in its 1H NMR spectrum, at d 1.4 (9H) and d 2.0 (3H). Its IR spectrum shows a strong absorption band near 1740 cm-1. What is the structure for this compound? Show the correlation of the spectra with the molecular structure.
Following are IR and 1H-NMR spectra of compound D. The mass spectrum of compound D shows a molecular ion peak at m/z 136, a base peak at m/z 107, and other prominent peaks at m/z 118 and 59.
Q.) Propose structural formulas for ions in the mass spectrum at m/z 118, 107, and 59.
Chapter 14 Solutions
Organic Chemistry
Ch. 14 - PRACTICE PROBLEM 14.1 Provide a name for each of...Ch. 14 - Prob. 2PPCh. 14 - Prob. 3PPCh. 14 - Practice Problem 14.4 Apply the polygon-and-circle...Ch. 14 - Practice Problem 14.5 Apply the polygon-and-circle...Ch. 14 - Practice Problem 14.6 1,3,5-Cycloheptatriene is...Ch. 14 - Prob. 7PPCh. 14 - Prob. 8PPCh. 14 - Practice Problem 14.9 In 1967 R. Breslow (of...Ch. 14 - Prob. 10PP
Ch. 14 - Practice Problem 14.11 In addition to a signal...Ch. 14 - PRACTICE PROBLEM 14.12
Azulene has an appreciable...Ch. 14 - Practice Problem 14.13 (a) The -Sh group is...Ch. 14 - Practice Problem 14.14
Explain how NMR...Ch. 14 - PRACTICE PROBLEM 14.15 Four benzenoid compounds,...Ch. 14 - Prob. 16PCh. 14 - Write structural formulas and give acceptable...Ch. 14 - Prob. 18PCh. 14 - Prob. 19PCh. 14 - Prob. 20PCh. 14 - Which of the hydrogen atoms shown below is more...Ch. 14 - 14.22 The rings below are joined by a double bond...Ch. 14 - Prob. 23PCh. 14 - 14.24 (a) In 1960 T. Katz (Columbia University)...Ch. 14 - Prob. 25PCh. 14 - Prob. 26PCh. 14 - 14.27 5-Chloro-1,3-cyclopentadiene (below)...Ch. 14 - Prob. 28PCh. 14 - Furan possesses less aromatic character than...Ch. 14 - 14.30 For each of the pairs below, predict...Ch. 14 - Assign structures to each of the compounds A, B,...Ch. 14 - Prob. 32PCh. 14 - Give a structure for compound F that is consistent...Ch. 14 - Prob. 34PCh. 14 - Prob. 35PCh. 14 - The IR and 1H NMR spectra for compound X(C8H10)...Ch. 14 - Prob. 37PCh. 14 - Prob. 38PCh. 14 - 14.39 Given the following information, predict the...Ch. 14 - Consider these reactions: The intermediate A is a...Ch. 14 - Prob. 41PCh. 14 - Compound E has the spectral features given below....Ch. 14 - Draw all of the molecular orbitals for...Ch. 14 - Prob. 1LGPCh. 14 - Prob. 2LGPCh. 14 - 3. The NMR signals for the aromatic hydrogens of...Ch. 14 - Prob. 4LGPCh. 14 - Prob. 5LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Why isn't FeBr3 used as a catalyst in the first step of the synthesis of 1,3,5-tribromobenzene?
Organic Chemistry (8th Edition)
17.24 A buffer is prepared by adding 10.0 g of ammonium chloride (NH4CI) to 250 mL of 1.00 M NH3 solution.
What...
Chemistry: The Central Science (13th Edition)
The heat of solution of ammonia in water at 1 atm is Hs(25C,r=2molH2O/molNH3)=78.2kJ/mol Calculate the enthalpy...
Elementary Principles of Chemical Processes, Binder Ready Version
a. Draw the most stable conformation of pentane for rotation about the C-2C-3 bond. b. Draw the least stable co...
Essential Organic Chemistry (3rd Edition)
a. Draw the mechanism for the following reaction if it a involves specific-base catalysis. b. Draw the mechanis...
Organic Chemistry
a. How does increasing the temperature increase the rate of a chemical reaction? b. How does increasing the amo...
General, Organic, and Biological Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardKetones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm-1 and M+=74 in the mass spectrum?arrow_forwardThe treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formula C5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forward
- The treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formulaC5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forwardCompound B of molecular formula C9H19N shows a noteworthy infrared absorption at 3300 cm-1. Its 1H-NMR spectrum shows three singlets – δ 1.0 (6H), 1.1 (12H), 1.4 (1H) ppm. Its 13C-NMR spectrum has four signals – δ 25, 28, 41, 64 ppm. Suggest a structure for this compound. Please show work.arrow_forwardFollowing are 1H-NMR spectra for compounds B (C6H12O2) and C (C6H10O). Upon warming in dilute acid, compound B is converted to compound C. Deduce the structural formulas for compounds B and C.arrow_forward
- Propose a structure for each of the following two isomers with formula C6H14 given their 1H-NMR spectra. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (t, 9H), 1.22 (q, 2H) ppmarrow_forwardAn unknown compound A (molecular formula C7H14O) was treated with NaBH4 in CH3OH to form compound B (molecular formula C7H16O). Compound A has a strong absorption in its IR spectrum at 1716 cm−1. Compound B has a strong absorption in its IR spectrum at 3600−3200 cm−1. The 1H NMR spectra of A and B are given. What are the structures of A and B?arrow_forwardTwo compounds with the molecular formula C5H10O have the following 1H and 13C NMR data. Both compounds have a strong IR absorption band in the 1710–1740-cm_1 region. Elucidate the structure of these two compounds and interpret the spectra. (10) (a) 1H NMR: d 2.55 (septet, 1H), 2.10 (singlet, 3H), 1.05 (doublet, 6H); 13C NMR: d 212.6, 41.5, 27.2, 17.8 (b) 1H NMR: d 2.38 (triplet, 2H), 2.10 (singlet, 3H), 1.57 (sextet, 2H), 0.88 (triplet, 3H); 13C NMR: d 209.0, 45.5, 29.5, 17.0, 13.2arrow_forward
- Compound B of molecular formula C9H19N shows a noteworthy infrared absorption at 3300 cm-1. Its 1H-NMR spectrum shows three singlets – δ 1.0 (6H), 1.1 (12H), 1.4 (1H) ppm. Its 13C-NMR spectrum has four signals – δ 25, 28, 41, 64 ppm. Suggest a structure for this compound.arrow_forwardCompounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forwardConsider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major produces) formed when this compound is treated with one equivalent of Br2?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY