
Loose Leaf Student Solutions Manual Organic Chemistry
10th Edition
ISBN: 9781259968907
Author: Francis Carey, Robert Giuliano
Publisher: McGraw-Hill Education (edition 10)
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 50P
Figure
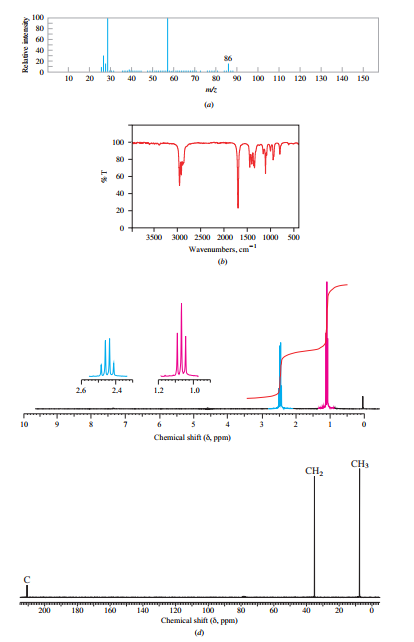
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which spectroscopic tool would be best for distinguishing a sample of 1,2-dichloropropane from 1,2-dibromopropane?
NMR spectroscopy
IR spectroscopy
UV/vis spectroscopy
Mass Spectrometry
Account for the formation of the base peaks in these mass spectra.
Q.) Isobutylmethylamine, m/z 44
What are the major IR bands, 1H-nmr signals, And the m/z of the following compounds
dibenzal acetone
Chapter 14 Solutions
Loose Leaf Student Solutions Manual Organic Chemistry
Ch. 14.3 - Prob. 1PCh. 14.3 - Prob. 2PCh. 14.4 - The 1H NMR signal for bromoform (CHBr3) appears at...Ch. 14.5 - identify the most shielded and least shielded...Ch. 14.5 - (a) Assign the chemical shifts 1.6, 2.2, and 4.8...Ch. 14.5 - Assign the chemical shifts 1.1, 1.7, 2.0, and 2.3...Ch. 14.5 - Assign the chemical shifts 1.6, 4.0, 7.5, 8.2, and...Ch. 14.6 - The 300-MHz 1H NMR spectrum of 1,4-dimethylbenzene...Ch. 14.6 - Prob. 9PCh. 14.6 - How many signals would you expect to find in the...
Ch. 14.7 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.8 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.11 - Prob. 13PCh. 14.11 - Prob. 14PCh. 14.12 - Hydrogen bonding between the oxygen of dimethyl...Ch. 14.14 - Prob. 16PCh. 14.15 - The 13C NMR spectrum of 1-bromo-3-chloropropane...Ch. 14.15 - Consider carbons x, y, and z in p-methylanisole....Ch. 14.15 - Prob. 19PCh. 14.16 - To which of the compounds of Problem 14.16 does...Ch. 14.18 - DEPT spectra for a compound with the formula...Ch. 14.20 - Vibrational frequencies are sensitive to isotopic...Ch. 14.21 - Prob. 23PCh. 14.22 - Prob. 24PCh. 14.23 - Prob. 25PCh. 14.23 - Which one of the C5H8 isomers shown has its max at...Ch. 14.24 - Knowing what to look for with respect to isotopic...Ch. 14.24 - The base peak appears at m/z105 for one of the...Ch. 14.24 - Mass spectra of 1-bromo-4-propylbenzene and...Ch. 14.25 - Prob. 30PCh. 14 - Each of the following compounds is characterized...Ch. 14 - Deduce the structure of each of the following...Ch. 14 - From among the isomeric compounds of molecular...Ch. 14 - The H1NMR spectrum of fluorene has signals at 3.8...Ch. 14 - Prob. 35PCh. 14 - H1NMR spectra of four isomeric alcohols with...Ch. 14 - Prob. 37PCh. 14 - We noted in Section 14.13 that an NMR spectrum is...Ch. 14 - Identify each of the C4H10O isomers on the basis...Ch. 14 - A compound (C3H7ClO2) exhibited three peaks in its...Ch. 14 - Label nonequivalent carbons in the following...Ch. 14 - Compounds A and B are isomers of molecular formula...Ch. 14 - C13 NMR spectra for four isomeric alkyl bromides...Ch. 14 - Prob. 44PCh. 14 - Prob. 45PCh. 14 - Identify the C3H5Br isomers on the basis of the...Ch. 14 - Prob. 47PCh. 14 - A compound (C8H10O) has the IR and H1NMR spectra...Ch. 14 - Deduce the structure of a compound having the...Ch. 14 - Figure 14.53 presents IR, H1NMR, C13NMR and mass...Ch. 14 - H1NMR, C13NMR, IR, and mass spectra are shown for...Ch. 14 - 1H NMR and IR spectra for a compound with the...Ch. 14 - FriedelCraftsalkylation of benzene with...Ch. 14 - Prob. 54DSPCh. 14 - Prob. 55DSPCh. 14 - Prob. 56DSPCh. 14 - Prob. 57DSPCh. 14 - Prob. 58DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Treatment of 3,4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how you would use 1H and 13CNMR spectroscopy to help identify them. How would you use UV spectroscopy?arrow_forwardWhat are the major IR bands, 1H-nmr signals, And the m/z of the following compounds benzaldehydearrow_forwardWhat are the major IR bands, 1H-nmr signals, And the m/z of the following compounds triphenylmethanolarrow_forward
- which peaks can be expected when 5-methylhexane-2,4-dione is analyzed by mass spectrometry?arrow_forwardCircle the solvents from the following list that can be used with 1H NMR spectroscopy that do not interfere with the spectrum. carbon tetrachloride chloroform benzene-d6 hexachloroacetone acetonitrile acetone methylene chloride D2O DMF (dimethylformamide)arrow_forwardFigures 2 to 4 show the IR, 1H NMR and 13C NMR spectra of a compound with formula C3H5N 2a) Identify bands A and B in the IR spectrumarrow_forward
- For this experiment depicted in the picture, Interpret the most important absorbances in the IR spectrum of the essential oil eugenol.arrow_forward9) What would be the difference between the 4 alcohols in IR? They all show O-H, C-H sp3, and C-H aromatic bonds. The only way to tell which one is used would be by comparing the fingerprint regions to published IR spectra of each alcohol. Alcohol 2 has an aldehyde C-H group that could be seen between 2700-2800 and 2800-2900 cm-1 Only alcohol 3 will show O-H bond around 3300 cm-1 Only alcohol 1 will show C=C aromatic bonds around 1600 cm-1 10) Assuming 5.0 mL of alcohol 2 was used and 2.2 g of the product was isolated, what is the percent yield for this reaction? The product has a molar mass of 198.00 g/mol. 11) How could the percent yield be increased for this reaction? Add NaOH to neutralize the product as it forms Add a catalyst such as H2SO4 to help speed up the reaction Add a catalyst such as HCl to help speed up the reaction Cool the reaction on ice to increase the rate of reaction 12) Is there another way that the product of this reaction…arrow_forwardA couple questions using H-NMR and IR spectroscopy.arrow_forward
- Predict the structure of the compound using the information below. Kindly show what information are obtained for each spectra which may help in predicting the structure. From IR Spectra: See the attached picture From Mass Spectra M+ = 134 base peak = 105 peaks at 77 and 51 From Proton and Carbon NMR Spectra See attached picture.arrow_forwardIn the lab, you are given 3-nitroacetophenone. If you reduce this with NaBH4, you will receive 3-nitrophenol as the final product. On an IR spectroscopy, what would be the IR peaks of both 3-nitroacetophenone and 3-nitrophenol?arrow_forwardthe mass spectrum, IR and 13 C and 1 HNMR spectra for an unknown organic molecule. Determine the structure ofthe molecule.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY