
Organic Chemistry Study Guide and Solutions Manual, Books a la Carte Edition (8th Edition)
8th Edition
ISBN: 9780134649771
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 79P
Identify each of the following compounds from its mass spectrum, 1R spectrum, and 1H NMR spectrum:
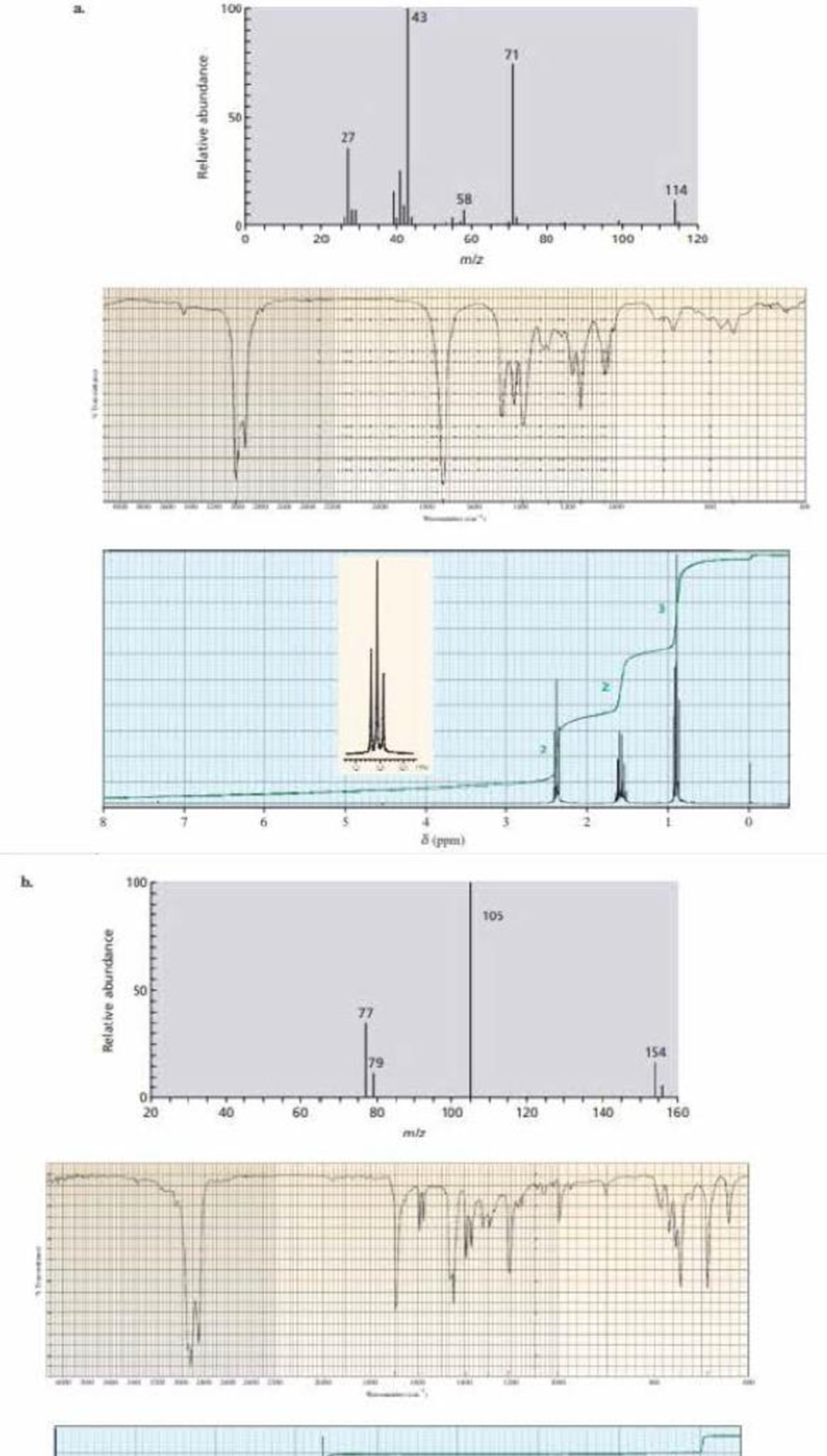
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify each of the following compounds from its molecular formula and its 1H NMR spectrum:
Identify the compound with molecular formula C4H9BrO that gives the following 1H NMR spectrum.
Identify each compound below from its molecular formula and its 13C NMR spectrum.
Chapter 14 Solutions
Organic Chemistry Study Guide and Solutions Manual, Books a la Carte Edition (8th Edition)
Ch. 14.1 - Prob. 1PCh. 14.1 - Prob. 2PCh. 14.4 - How many signals would you expect to see in the 1H...Ch. 14.4 - How many signals would you expect to see in the 1H...Ch. 14.4 - How could you distinguish the 1H NMR spectra of...Ch. 14.4 - Draw an isomer of dichlorocyclopropane that gives...Ch. 14.5 - Prob. 7PCh. 14.5 - Prob. 8PCh. 14.5 - Prob. 9PCh. 14.5 - Where would you expect to find the 1H NMR signal...
Ch. 14.6 - Prob. 11PCh. 14.7 - Prob. 12PCh. 14.7 - Prob. 13PCh. 14.7 - Without referring to Table 14.1, label the proton...Ch. 14.8 - [18]-Annulene shows two signals in its 1H NMR...Ch. 14.9 - How would integration distinguish the 1H NMR...Ch. 14.9 - Which of the following compounds is responsible...Ch. 14.10 - Prob. 19PCh. 14.10 - Explain how the following compounds, each with the...Ch. 14.10 - The 1H NMR spectra of two carboxylic acids with...Ch. 14.11 - Draw a diagram like the one shown in Figure 14.12...Ch. 14.12 - Indicate the number of signals and the...Ch. 14.12 - Explain the relative chemical shifts of the...Ch. 14.12 - How can their 1H NMR spectra distinguish the...Ch. 14.12 - Identify each compound from its molecular formula...Ch. 14.12 - Predict the splitting patterns for the signals...Ch. 14.12 - Describe the 1H NMR spectrum you would expect for...Ch. 14.12 - Propose structures that are consistent with the...Ch. 14.13 - Prob. 30PCh. 14.13 - Identify the compound with molecular formula...Ch. 14.14 - Prob. 32PCh. 14.15 - a. For the following compounds, which pairs of...Ch. 14.15 - How would the 1H NMR spectra for the four...Ch. 14.17 - Explain why the chemical shift of the OH proton of...Ch. 14.17 - Prob. 38PCh. 14.17 - Prob. 39PCh. 14.17 - Prob. 40PCh. 14.20 - Answer the following questions for each compound:...Ch. 14.20 - Prob. 42PCh. 14.20 - How can 1,2-, 1,3-, and 1,4-dinitrobenzene be...Ch. 14.20 - Identify each compound below from its molecular...Ch. 14.22 - Prob. 45PCh. 14.22 - What does cross peak X in Figure 14.34 tell you?Ch. 14 - Prob. 47PCh. 14 - Draw a spitting diagram for the Hb proton and give...Ch. 14 - Label each set of chemically equivalent protons,...Ch. 14 - Determine the ratios of the chemically...Ch. 14 - How can 1H NMR distinguish between the compounds...Ch. 14 - Prob. 52PCh. 14 - Match each of the 1H NMR spectra with one of the...Ch. 14 - The 1H NMR spectra of three isomers with molecular...Ch. 14 - Prob. 55PCh. 14 - Prob. 56PCh. 14 - Compound A, with molecular formula C4H9Cl, shows...Ch. 14 - Would it be better to use 1H NMR or 13C NMR...Ch. 14 - There are four esters with molecular formula...Ch. 14 - Identify the compound with molecular formula C6H14...Ch. 14 - An alkyl halide reacts with an alkoxide ion to...Ch. 14 - The 1H NMR spectra of three isomers with molecular...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Prob. 65PCh. 14 - How can the signals in the 6.5 to 8.1 ppm region...Ch. 14 - The 1H NMR spectra of two compounds, each with...Ch. 14 - Draw a splitting diagram for the Hb proton if Jbc...Ch. 14 - Sketch the following spectra that would be...Ch. 14 - How can 1H NMR be used to prove that the addition...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Dr. N. M. Arr was called in to help analyze the 1H...Ch. 14 - Calculate the amount of energy (in calories)...Ch. 14 - The following 1H NMR spectra are four compounds,...Ch. 14 - When compound A (C5H12O) is treated with HBr, it...Ch. 14 - Identity the compound with molecular formula...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Prob. 78PCh. 14 - Identify each of the following compounds from its...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A compound of unknown structure gave the following spectroscopic data: Mass spectrum: M+=88.1 IR: 3600 cm-1 1ΗNMR: 1.4 δ (2 H, quartet, J=7 Hz); 1.2 δ (6H, singlet): 1.0 δ (1 H, singlet); 0.9 δ (3 H, triplet, J=7 Hz) 13CNMR: 74, 35, 27, 25 δ (a) Assuming that the compound contains C and H but may or may not contain O, give three possible molecular formulas (b) How many protons (H) does the compound contain? (c) What functional groups(s) does the compound contain? (d) How many carbons does the compound contain? (e) What is the molecular formula of the compound? (f) What is the structure of the compound? (g) Assign peaks in the molecule’s 1HNMR spectrum corresponding to specific protons.arrow_forwardCompound F, a hydrocarbon with M+=96 in its mass spectrum, undergoes reaction with HBr to yield compound G. Propose structures for F and G, whose 13C NMR spectral data are given below. Compound F Broadband-decoupled 13C NMR: 27.6, 29.3, 32.2, 132.4 DEPT-90: 132.4 DEPT-135: positive peak at 132.4 ; negative peaks at 27.6, 29.3, 32.2 Compound G Broadband-decoupled 13C NMR: 25.1, 27.7, 39.9, 56.0 DEPT-90: 56.0 DEPT-135: positive peak at 56.0 ; negative peaks at 25.1, 27.7, 39.9arrow_forwardIdentify the compound with molecular formula C3H7NO responsible for the 1H NMR spectrum shown here.arrow_forward
- draw the structure of the nmr (ethyl acetate) then circle the protons on the structures and correlate the 1H's to the resonances (peaks) on the 1H NMR spectra.arrow_forwardIdentify the compound with molecular formula C6H14 that is responsible for the following 1H NMR spectrum:arrow_forward1H NMR spectrum is shown below. Is it an ester found in wintergreen oil or an ester that is commonly used as a pain reliever. Both esters can be made from salicylic acid. Based on this information, determine the structure/identity of the ester corresponding to Spectrum 1.arrow_forward
- Treatment of butan-2-one (CH3COCH2CH3) with strong base followed by CH3I forms a compound Q, which gives a molecular ion in its mass spectrum at 86. The IR (> 1500 cm−1 only) and 1H NMR spectrum of Q are given below. What is the structure of Q?arrow_forwardDraw the structure of ethonic acid, then circle the protons on the structures and correlate the 1H's to the resonances (peaks) on the 1H NMR spectra.arrow_forwardcompound gives only one signal in its 1H-NMR spectrum. Propose a structural formula for compound. Q.) C6H12arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY