
Concept explainers
The chapter sections to review are shown in parentheses at the end of each problem.
14.87 Determine if each of the following diagrams represents a strong acid or a weak acid. The acid has the formula HX. (14.3)
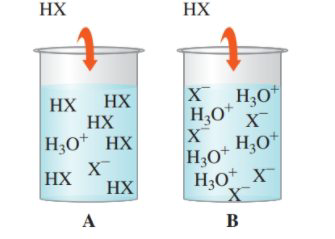
Interpretation:
To identify the strong and weak acids, given the diagrams with the ion species.


Concept introduction:
- Weak acids will not dissociate completely
- Strong acid will dissociate 100% in solution.
To determine: The weak and strong acids
Answer to Problem 87UTC
Solution: weak acid is A; strong acid is B
Explanation of Solution
An acid is said to be a strong acid if it will be 100% dissociated into ions in the solution. The acid will donate all of its acidic protons, and therefore, will have a higher hydronium ion concentration in solution after dissolution. The pH is likely to be lower.
An acid is said to be a weak acid if it will NOT be 100% dissociated into ions in the solution. The acid will not donate all its acidic protons, and therefore, will form equilibrium between ions and acid. The pH is likely to be higher.
Since, the number of ions (
Weak acids and strong acids can be identified if the dissociation factor is given.
Want to see more full solutions like this?
Chapter 14 Solutions
EBK BASIC CHEMISTRY
- Write a balanced equation for the double-replacement neutralization reaction described, using the smallest possible integer coefficients.A reaction occurs when aqueous solutions of carbonic acid and sodium hydroxide are combined. Assume excess base.arrow_forwardBaking soda (sodium bicarbonate, NaHCO3) reacts withacids in foods to form carbonic acid (H2CO3), which inturn decomposes to water and carbon dioxide gas. In a cake batter, the CO2(g) forms bubbles and causes the caketo rise. (a) A rule of thumb in baking is that 1/2 teaspoon ofbaking soda is neutralized by one cup of sour milk. The acidcomponent in sour milk is lactic acid, CH3CH(OH)COOH.Write the chemical equation for this neutralization reaction.(b) The density of baking soda is 2.16 g/cm3. Calculatethe concentration of lactic acid in one cup of sour milk(assuming the rule of thumb applies), in units of mol/L.(One cup = 236.6 mL = 48 teaspoons). (c) If 1/2 teaspoonof baking soda is indeed completely neutralized by the lactic acid in sour milk, calculate the volume of carbon dioxidegas that would be produced at 1 atm pressure, in anoven set to 350 °F.arrow_forwardAn acid has a Ka of 4.53 * 10-6 in water. What is its Keq for reaction with water in a dilute solution? ([H2O] = 55.5 M)arrow_forward
- Calculate the [H3O+] value of each aqueous solution. aspirin with [OH−]=1.2×10−11M .Express your answer using two significant figures.arrow_forwardA base is found to have a Kb = 6.38 at 25 °C. What is the pKa (to two decimal places) of the conjugate acid?arrow_forwardMagnesium reacts with sulfuric acid in a single displacement reaction. How manymilligrams of hydrogen gas can be produced from the reaction of 36.00 mL of a sulfuricacid solution with a pH of 1.731 and excess magnesium? answer in milligrams!!!arrow_forward
- decreasing tendency to donate a protonarrow_forwardhow do you enter this into a calculator to get 5.8arrow_forwardVinegar contains acetic acid and it's used in salad dressings.what if acetic acid was a strong acid instead of a weak acid?would it be safe to use vinegar as a salad dressing?arrow_forward
- 1. Standard acids are commonly standardized using sodium carbonate, while standard bases are standardized by potassium hydrogen phthalate. Select one: True False 2. Which of the following is not used as a standard acid because it could oxidize the visual indicators? a. Perchloric acid b. Sulfuric acid c. Nitric acid d. Hydrochloric acidarrow_forwardWhat does the value of Kw say about the extent to which water molecule react with themselves? a. Water molecules react with themselves at constant rate, never increasing or decreasing. b. When an acid is added to water, the value of Kw will decrease. c. Because the value of Kw is so small, the extent to which water ionizes is quite large. d. Because the value of Kw is so small, the extent to which water ionizes is quite small.arrow_forward, Neutralization of acid lake with calcium carbonate: How much limestone, CaCO3, completely neutralizes a 4.3 billion liter lake with a pH of 5.5?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


