
Concept explainers
(a)
Interpretation:
The given molecule has to be identified as an acid, an
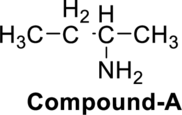
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
There are different types of
(b)
Interpretation:
The given molecule has to be identified as an acid, an amine and both or neither.
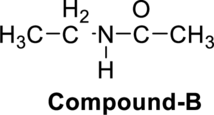
Concept Introduction:
Refer to part (a)
(c)
Interpretation:
The given molecule has to be identified as an acid, an amine and both or neither.
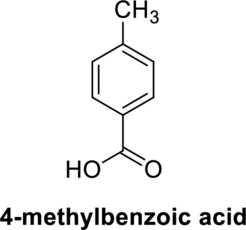
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The given molecule has to be identified as an acid, an amine and both or neither.
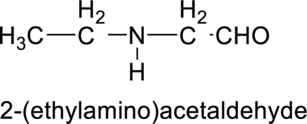
Concept Introduction:
Refer to part (a).
(e)
Interpretation:
The given molecule has to be identified as an acid, an amine and both or neither.
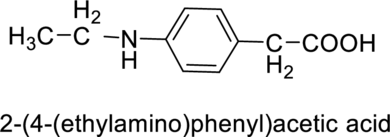
Concept Introduction:
Refer to part (a)
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
OWLV2 FOR MOORE/STANITSKI'S CHEMISTRY:
- TRUE OR FALSE (a) There are three amines with the molecular formula C3H9N. (b) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (c) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (d) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (e) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (f) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forwardDraw the skeletal ("line") structure of a secondary amine with 4 carbon atoms, and no double or triple bonds.arrow_forward___are considered weak bases as they are capable of removing H+ ions from solution. Meanwhile___ are considered weak acids as they can donat H+ ions into the solution. is it amines and carboxylic acids?arrow_forward
- Which of the following examples are acid derivatives? A ) Amines B ) amides C ) ester D ) aldehydes E ) acyl chloridearrow_forwardIs this an ether, ester, carboxylic acid, aldehyde, or amide?arrow_forwardExplain why it is impossible to form an amide by the condensation of a tertiary amine with a carboxylic acid.arrow_forward
- Write the common name for each amine. HỌC—N—CH,CH3 H₂C-CH₂-CH₂-CH₂ Ethylbutylamine common name: Incorrect HC—CH,—N—CH,—CHy CH₂ CH3 diethylethylamine common name: Incorrect These compounds are tertiary amines.arrow_forwardWrite the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forward(i) Draw the structure of any amine and give the IUPAC name of that amine. (i) Classify the amine in your answer provided in (i) above (iii) Draw the structure of ethyl butanoate and name the functional group. (iv) Give the IUPAC name of the following compound and name the functional group:arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

