
The decomposition of Hl(g) is represented by the equation
Hl(g) is introduce into five identical 400 cm2 glass bulbs, and the five bulbs are maintained at 623 K. Each bulb is opened after a period of time and analyzed for
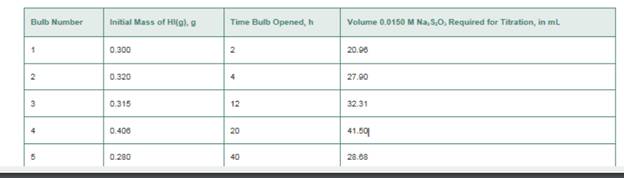
Data for this experiment are provided in the table. What is the value of
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
- For the decomposition of Ag2O: 2Ag2O(s)4Ag(s)+O2(g)(a) Obtain an expression for G as a function of temperature. Prepare a table of G values at 100 K intervals between 100 K and 500 K. b) Calculate the temperature at which G =0.arrow_forward5.12. True or false: If all the partial pressures of reactants and products drop by half, the value of Q drops by half. Give an example of a chemical reaction to support your answer.arrow_forwardMonochloroethane (C2H5Cl) can be produced by the direct reaction of ethane gas (C2H6) with chlorine gas or by the reaction of ethylene gas (C2H4) with hydrogen chloride gas. The second reaction gives almost a 100% yield of pure C2H5Cl at a rapid rate without catalysis. The first method requires light as an energy source or the reaction would not occur. Yet G for the first reaction is considerably more negative than G for the second reaction. Explain how this can be so.arrow_forward
- A. For a certain reaction, Kc = 4.48×106 and kf= 4.10×10−2 M−2⋅s-1 . Calculate the value of the reverse rate constant, kr, given that the reverse reaction is of the same molecularity as the forward reaction. Express your answer with the appropriate units. Include explicit multiplication within units, for example to enter M−2⋅s−1nclude ⋅⋅ (multiplication dot) between each measurement. B. For a different reaction, Kc = 97.7, kf=3.00×10^3s−1 and kf=3.00×10^3s−1 , and kr= 30.7 s−1. Adding a catalyst increases the forward rate constant to 1.37×106 s−1s−1 . What is the new value of the reverse reaction constant, kr, after adding catalyst? Express your answer with the appropriate units. Include explicit multiplication within units, for example to enter M−2⋅s−1 include ⋅ (multiplication dot) between each measurement.arrow_forwardFor a certain reaction, Kc�� = 9.80×10−10 and kf=�f= 2.59×10−5 M−2⋅s−1�−2⋅s−1 . Calculate the value of the reverse rate constant, kr�r, given that the reverse reaction is of the same molecularity as the forward reaction. Express your answer with the appropriate units. Include explicit multiplication within units, for example to enter M−2⋅s−1�−2⋅�−1 include ⋅⋅ (multiplication dot) between each measurement. View Available Hint(s) For a different reaction, Kc�� = 5.40×102, kf=8.96×103s−1�f=8.96×103s−1 , and kr=�r= 16.6 s−1s−1 . Adding a catalyst increases the forward rate constant to 2.05×106 s−1s−1 . What is the new value of the reverse reaction constant, kr�r, after adding catalyst? Express your answer with the appropriate units. Include explicit multiplication within units, for example to enter M−2⋅s−1�−2⋅�−1 include ⋅⋅ (multiplication dot) between each measurement. View Available Hint(s)arrow_forwardCalculate the equilibrium constant reaction CO(g) + H2(g) H2CO(g), given that for the production of liquid methanal, ∆rG −◦ = +28.95 kJ mol−1 at 298 K and that the vapor pressure of methanal is 1500 torr at the given temperature.arrow_forward
- At 1000 K, Kp for the reaction given below is equal to 3.42: 2 SO2(g) + O2(g) ⇌ 2 SO3(g) In a reaction mixture at 1000 K, it was determined that there was 0.20 mol of SO2, 0.30 mol of O2 and 0.55 mol of SO3, with a total pressure of 1.5 x 105 Pa. What is the value of Qp for the system? a. 0.0568 b. 17.6 c. 5.68 x 103 d. 1.76 x 10-4 e. 11.7arrow_forwardFor the reaction 2CH4(g)⇌C2H2(g)+3H2(g)2CH4(g)⇌C2H2(g)+3H2(g) KcKcK_c = 0.155 at 1611 ∘C∘C . What is KpKpK_p for the reaction at this temperature? Express your answer numerically.arrow_forwardCO(g)+2H2(g)⇌CH3OH(g) K = 2.18×102 at 340 K Calculate ΔrG for the reaction at 340 K in STANDARD CONDITIONS please Express your answer using three significant figures and include the appropriate units.arrow_forward
- Consider the equilibrium system described by the chemical reaction below, which has a value of Kc equal to 4.10 × 10⁻⁴ at a high temperature. If 0.20 mol N₂ and 0.15 mol O₂ react in a 1.0 L vessel, what will the equilibrium concentration of O₂ be? N₂(g) + O₂(g) ⇌ 2 NO(g) A )Set up the expression for Qc and then evaluate it to determine the direction of the reaction. Do not combine or simplify terms. Qc = _____/_____ = _____ Based on the given values and your value for Qc, set up ICE table in order to determine the unknown. Based on your ICE table, set up the expression for Kc in order to determine the unknown. Do not combine or simplify terms. Kc = (________) / (_______)= 4.10 × 10⁻⁴ Based on your ICE table and expression for Kc, solve for the concentration of O₂ at equilibrium. [O₂]eq = _____Marrow_forwardThe value of ΔG° at 25 °C for the decomposition of phosphorous trichloride into its constituent elements, 2PCl3 (g) → P2 (g) + 3Cl2 (g)is ________ kJ/mol.A) -539.2B) +539.2C) -642.9D) +642.9E) -373.3arrow_forwardA 10.0 LL reaction vessel contains 4.40 gg of CO2(g)CO2(g), 2.00 gg of C(s)C(s), and 6.785 gg of CO(g)CO(g). In the presence of graphite (solid carbon), carbon dioxide reacts as follows to form carbon monoxide: CO2(g)+C(s)⇌2CO(g), Kc=5.87×10−2CO2(g)+C(s)⇌2CO(g), Kc=5.87×10−2 at 1200K In which direction does the reaction proceed to reach equilibrium? a. Q=K, the reaction system is at equilibrium. b. Q>K, reaction goes to the right c. Q<K, the reaction system proceeds to the right. d. Q>K, the reaction system proceeds to the left.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,





