
Interpretation:
The product of the given reaction has to be shown.
Concept Introduction:
Simmons-Smith reaction: This is ultrasonication improve the rate of formation of these organic zinc compounds, as with many organometallic reactions occurring at a surface condition.
Example: The substance of a carbenoid a carbine like substance that converters
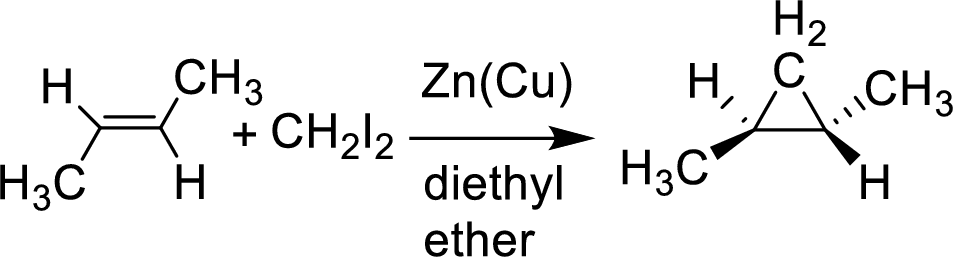
Addition Reaction:
- It is a type of reaction in which two reactants adding together to form a single product.
- It can be said as a reverse reaction of elimination reaction.
- It is a characteristic reaction of
alkane .
Hydrogenation reaction is an addition reaction in which addition of hydrogen to an unsaturated molecule occurs
Nucleophiles: A nucleophile is a more reactant species that affords a pair of electrons to the electrophile or electrophilic center and forms a new covalent bond. The carbon or other hetero atom in a molecule which is bearing negative charge or lone pair of electron is called as nucleophiles.
Regiochemistry: if the
Stereochemistry: when the attack takes place at the chiral center, inversion of configuration is observed. Only the center being attacked undergoes an inversion of configuration.
Stereospecific reaction: reaction undergoes from a stereoisomer to a unique stereo isomeric product.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EP ORGANIC CHEMISTRY-OWL V2 ACCESS
- A student proposes the following reaction mechanism for the reaction in Model 6. Which step inthis mechanism is least favorable? Explain your reasoning.arrow_forwardDraw the product of the synthesis shown above.arrow_forwardThe following molecules are subject to substitution (SN1 or SN2) reaction conditions. a) Identify if the leaving group (-Br) is attached to a 3°, 2°, 1°, or methyl Carbon. b) Rank the molecule with respect to their SN1 reactivity, with 1 being the fastest and 4 being the slowest. c) Rank the molecule with respect to their SN2 reactivity, with 1 being the fastest and 4 being the slowest. Br Br CH3B Br a. type of Carbon on C-Br b. SN1 reactivity (1 fastest, 4 slowest) c. SN2 reactivity (1 fastest, 4 slowest)arrow_forward
- Draw the product of the reaction shown.arrow_forwardComplete the following multi-step synthesis.arrow_forwardDraw the major organic product for the reaction shown. The starting material is a 5 carbon ring with a hydroxy substituent. This reacts with SOCl2 and pyridine to form the product.arrow_forward
- Draw the major product of this SN1 reaction. Ignore any inorganic byproducts.arrow_forwardComplete the following reactions in sequential order. Show the product after each step and put a box around the final product. (Show intermediate structure after each reagent)arrow_forwardDraw the product and mechanism for the reaction below. Include all major resonance structures.arrow_forward
- The reaction shown below proceeds by both an SN1 and an SN2 mechanism. Draw the products of each process and use mechanistic arguments to account for any differences in the products formed. Ph 'Br SH Basearrow_forwardDraw the product of the following reaction. Omit stereochemistry from the product structure. NaCN HCl Click and drag to start drawing a structure.arrow_forwardShow how you would synthesize the target compound ne right from the starting compound on the left. Show reagents and conditions, and the structures of important intermediate compounds. Do not show any (arrow pushing) mechanism. Ignore stereochemistry in this question.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
