
Interpretation:
The delocalization energy of
Concept introduction:
The term conjugated dienes is used when the double bonds are present alternatively in a hydrocarbon chain. The mathematical function that describes the wave like behavior of an electron in a molecule is expressed by molecular orbital.
The delocalization energy for conjugated molecules is defined as the extra stability that a molecule attains by spreading its electron over the entire molecule. The delocalization of
Answer to Problem 15.2P
The delocalization energy of
Explanation of Solution
The delocalization energy of
The energy of ethylene bonding molecular orbital is
The formula for the calculation of
The bonding molecular orbital (BMOs) of ethylene contains two electrons.
The calculation of
The calculation of
The diagram for orbital interaction in
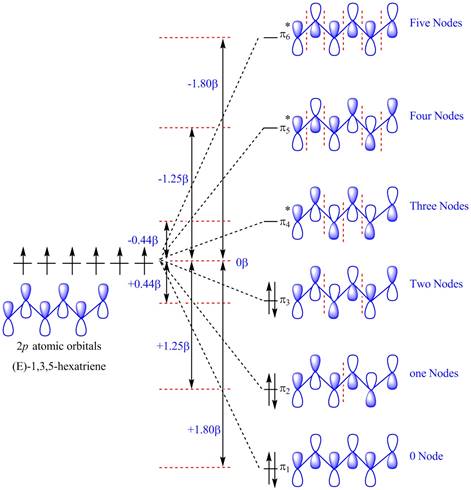
Figure 1
The
The energy of electrons in
The energy of electrons in
The energy of electrons in
Substitute the values of the energies of
Now, substitute the value from equation (3) and (5) in equation (1) in order to calculate the value of delocalization energy of the molecule
Now,
Therefore, the delocalization energy obtained for
The molecule
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry Study Guide and Solutions
- Draw the structure for (E,E) 3,5-octadienearrow_forward(a) Draw the molecular orbital picture for propa-1,2-diene, H2C=C=CH2. Hint: The three-dimensional geometry is shownin the chapter. (b) Draw the MO energy diagram for propa-1,2-diene. What is the HOMO? What is the LUMO?arrow_forward10) Consider the following reaction: Any: a) Shade the orbital lobes to provide the molecular orbital that is involved in the cyclization of this triene under the conditions provided. 888888 Δarrow_forward
- Draw structural formulas for the following compounds: (a) (2Z,4Z)-2,4-dibromo-3-methylhepta-2,4-diene (b) ((2E,4Z)-2-bromo-3,5-dichloro-4-methylhexa-2,4-dienearrow_forwardDistinguish Mechanistically between 1,4 and 1,2 isomers of 1,4-dienesarrow_forwardDraw the structure of (Z)-1-bromo-1-chloro-1-butene. • Consider E/Z stereochemistry of alkenes. • You do not have to explicitly draw H atoms. AAVII ? ChemDoodle [F Ⓡarrow_forward
- Draw a structure for (3E,5Z)-2,6-dimethyl-1,3,5,7-octatetraene.arrow_forwardq3carrow_forwardDraw a structural formula for the organic anion (i.e., do not include K+ or Nat) formed from the following reaction. Don't forget the charge on the ion. 0 сненансон CH3 CH4 +KOH →→ ? • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Include all nonzero formal charges. References to access important values if needed for this question. ? ChemDoodle Ⓡ Previousarrow_forward
- Draw the structure of (E)-2,3-dichloro-4-methyl-2- hexene. . Consider E/Z stereochemistry of alkenes. • You do not have to explicitly draw H atoms. **ELL [ ] در ?arrow_forwardDraw the skeletal (line-bond) structure of (1R,2S)-1-bromo-2-methylcyclohexanearrow_forwardTreating cyclohexene with Br₂ yields trans-1,2-dibromocyclohexane. Justify why the cis isomer is not formed. Br Br2 Brarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

