
Concept explainers
Problem Set
Anchoring Cells to the ECM. Animal cells attach to several different kinds of proteins within the ECM.
(a) Briefly explain how the various domains of the fibronectin molecule (see Figure 15-16) or the laminin molecule (see Figure 15-18) are important for their function.
(b) Historically, an important strategy for disrupting the adhesion of integrins to their ligands is by using a synthetic peptide that mimics the binding site on the ECM molecule to which the integrin attaches. In the case of fibronectin, the amino acid sequence is arginine-glycine-aspartate (when written using the single letter designation for each amino acid, this sequence becomes RGD). Explain why addition of such synthetic peptides would disrupt binding of cells to their normal substratum.
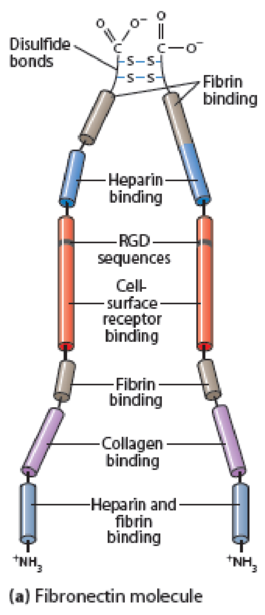
Figure 15-16 Fibronectin Structure. (a) A fibronectin molecule consists of two nearly identical polypeptide chains joined by two disulfide bonds near their carboxyl ends. Each polypeptide chain is folded into a series of domains linked by short, flexible segments. These domains have binding sites for ECM components or for specific receptors on the cell surface, including the tripeptide sequence RGD (arginine-glycine-aspartate), which is recognized by integrins. Besides the binding activities noted, fibronectin has binding sites for heparan sulfate, hyaluronate, and gangliosides (glycosphingolipids that contain sialic acid groups). (b) Myoblast cells on an ECM containing fibronectin in vitro, immunostained for fibronectin (green) and for DNA in the nucleus (blue).
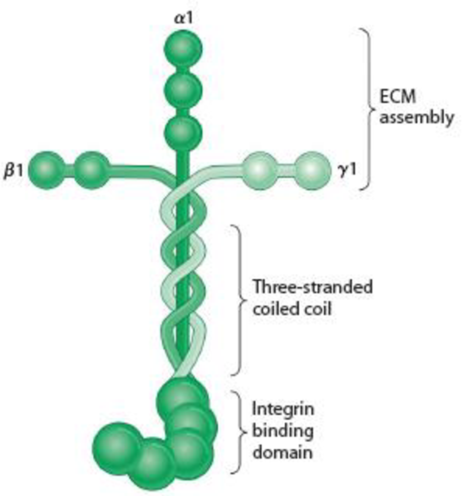
Figure 15-18 Laminin and the Basal Lamina. (a) A laminin molecule consists of three large polypeptides—α, β, and γ—joined by disulfide bonds into a crosslinked structure. A portion of the long arm consists of a three-stranded coil. Domains on the ends of the α chain are recognized by cell surface receptors; those at the ends of the two arms of the cross are specific for type IV collagen. The cross-arms also contain laminin-laminin binding sites, which enable laminin to form large aggregates. Laminin also contains binding sites for heparin, heparan sulfate, and entactin (not shown). (Adapted with permission from Macmillan Publishers Ltd: Fig. 1 from M. P. Marinkovich, “Laminin 332 in Squamous-Cell Carcinoma,” Nature Reviews Cancer 7:370-380. Copyright 2007.) (b) Laminin assembled into a basal lamina. Laminin associates with type IV collagen, perlecan, nidogen, and other components to form a mat of extracellular matrix. Cells attach to the basal lamina using integrins.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
WORLD OF CELL+MASTERING ACCESS >CUSTOM
- Multipass transmembrane proteins synthesized by ribosomes on the rough endoplasmic reticulum generally have which of the following arrangements of start-transfer and stop-transfer signals? multiple start signals and multiple stop signals (to allow multiple transmembrane regions) multiple start signals, but only one stop signal (to allow only one transmembrane region) only one start signal, but multiple stop signals (to allow only one transmembrane region) only one start signal, and only one stop signal (to allow only one transmembrane region) only one stop signal, and only one start signal (to allow only one transmembrane region)arrow_forwardDescrive in 2-3 sentences an in vitro experiment that differentiates between the Arp2/3 complex’ ability to associate with both the side of the ‘mother’ actin filament and with the barbed end of the ‘mother’ actin filament. Your experiment should take advantage of capping protein (binds and blocks barbed ends), and direct visualization of actin filaments.arrow_forwardIDENTIFICATION. HELLO, BASED ON MY PRPEVIOUS EXPERIENCE HERE, BARTLEBY EXPERTS CAN ANSWER UP TO 3 SUBPARTS. THANK YOU Identification. a)Carrier of the activated amino acids to the ribosomes for incorporation into the growing peptide chain. b)Molecule that attaches to myosin and actin during muscle contraction.arrow_forward
- performing corrections: Which coat protein is involved in transport of vesicles from the trans-Golgi network to the plasma membrane, endosomes, and lysosomes A. Clathrin B.COPI C.COPII D.Trench coat I had originally selected option C but got it wrong, what is the correct answer with explanation and how is the answer i selected incorrect?arrow_forwardQuestion:- What biological rationale can explain why there are so few variants observed at position 65 of the heme distal ligand and position 94 of the heme proximal ligand of myoglobin? Why does the number of variants differ between the two sites?arrow_forwardSIGNALS AND TARGETS. Listed below are sample polypeptides/proteins with their signal molecule/peptide. Answer the questions that follow. If you are asked to give the amino acid sequence, provide the sequence using the three-letter names of the amino acids (eg. ser-ala-met). Polymerase with H2N-...GMMTVPPKKKRVGMMTV...-COOH Provide the amino acid sequence of the signal peptide Where will this polypeptide be transported? What is the receptor of the signal sequence? What is the transport complex for this protein?arrow_forward
- Focal adhesion complex attachment to extracellular matrix molecules is mediated by: heterodimers of alpha-integrin and beta integrin homodimers of classical cafherins The force of gravity actin filamentsarrow_forwardQuestion:- Transport into the lumen of some organelle systems requires an unfolded protein structure, while for other import mechanisms, unfolding is not required, since proteins may be transported in the folded state across the membrane. How is it decided whether a freshly made protein will be imported into the lumen of the endoplasmic reticulum (ER)?arrow_forwardBriefly describe an in vitro experiment that differentiate between the Arp2/3 complex’ ability to associate with both the side of the ‘mother’ actin filament and with the barbed end of the ‘mother’ actin filament. Your experiment should take advantage of capping protein (binds and blocks barbed ends), and direct visualization of actin filaments. Draw diagrams of actin filaments (simple lines are okay) that indicate the possible outcomes of your experiment. describe your interpretation of the possible outcomes for the mechanism of Arp2/3 complex-mediated branch formationarrow_forward
- Question:- Biology 'Signal sequences and vesicular transport are important in the localisation and transport of proteins synthesized and processed in the Secretory pathway of eukaryotic cells.' Giving examples to illustrate your answer, describe the composition and relevnce of signal sequences and mechanisms involved in vesicular transport.arrow_forwardSIGNALS AND TARGETS. Listed below are sample polypeptides/proteins with their signal molecule/peptide. Answer the questions that follow. If you are asked to give the amino acid sequence, provide the sequence using the three-letter names of the amino acids (eg. ser-ala-met). Catalase with H2N-...KERINGKERIANGEKSAMSKL-COOH Provide the amino acid sequence of the signal peptide Where will this polypeptide be transported? (specify the compartment) What is the name of the specific receptor of this polypeptide? The receptor may also have what alternative function?arrow_forwardDescribe critical concentration (Cc)• Describe actin/microtubule polymerization• Contrast “T” and “D” forms of actin and microtubule subunits– Which nucleotide is bound to actin monomers in “T” and “D” forms?– Which nucleotide is bound to microtubule dimers in “T” and “D” forms? – Which subunit form, “T” or “D,” has higher affinity for polymerization? What are the implications of this on Cc(T) and Cc(D)?arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning

