
(a)
Interpretation:
The compound 1-pentene exhibit geometric isomerism or not has to be given and two isomers has to be drawn and named for the given structure.
Concept Introduction:
The structure of the compound is given by its systematic name.
To give the structure from the name of the compound, the root name has to be identified. The root name indicates the number of carbon atoms present in the longest chain.
Then the functional group (suffix) has to be identified. It indicates whether any functional groups are present in the compound, it also gives whether the compound is an
The prefix of the name indicates the branched groups and their positions on the carbon chain.
The name of the compound is in the form
Prefix + Root + Suffix
The geometric isomers are said to be the isomers which shows different orientation of groups around a double bond. The geometric isomers are also known as cis-trans isomers.
When two similar or higher priority groups are attached to the carbon on same side, it is said to be cis-isomer.
When two similar or higher priority groups are attached to the carbon on opposite sides, it is said to be trans-isomer.
To exhibit the geometric isomerism, a molecule should have double bonded carbon atoms (
(a)
Explanation of Solution
The given compound is 1-pentene.
The pentene contains five carbon atoms in chain. As the suffix is –ene, it belongs to alkene group and contains a double bond. The position of the double bond in 1-pentene is in between first and second carbon atoms. The structure of 1-pentene is given as
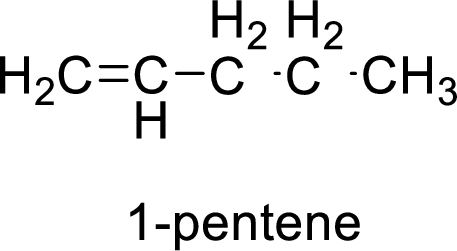
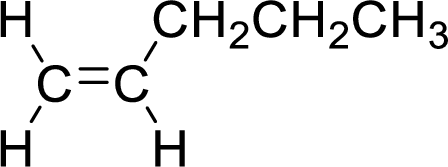
The given compound is 1-pentene. The given compound contains a double bond and each carbon in the double bond is bonded to two groups.
The geometric isomers are cis-isomer and trans-isomer. In cis-isomer, similar groups are attached to the carbon on same side and in trans-isomer, similar groups are attached to the carbon on opposite sides.
As 1-pentene contains two similar groups (
(b)
Interpretation:
The compound 2-pentene exhibit geometric isomerism or not has to be given and two isomers has to be drawn and named for the given structure.
Concept Introduction:
The structure of the compound is given by its systematic name.
To give the structure from the name of the compound, the root name has to be identified. The root name indicates the number of carbon atoms present in the longest chain.
Then the functional group (suffix) has to be identified. It indicates whether any functional groups are present in the compound, it also gives whether the compound is an alkane or alkene or alkyne.
The prefix of the name indicates the branched groups and their positions on the carbon chain.
The name of the compound is in the form
Prefix + Root + Suffix
The geometric isomers are said to be the isomers which shows different orientation of groups around a double bond. The geometric isomers are also known as cis-trans isomers.
When two similar or higher priority groups are attached to the carbon on same side, it is said to be cis-isomer.
When two similar or higher priority groups are attached to the carbon on opposite sides, it is said to be trans-isomer.
To exhibit the geometric isomerism, a molecule should have double bonded carbon atoms (
(b)
Explanation of Solution
The given compound is 2-pentene.
The pentene contains five carbon atoms in chain. As the suffix is –ene, it belongs to alkene group and contains a double bond. The position of the double bond in 2-pentene is in between second and third carbon atoms. The structure of 2-pentene is given as
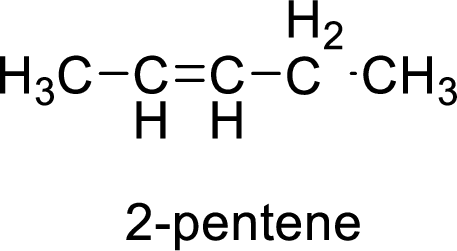
The given compound is 2-pentene. The given compound contains a double bond and each carbon in the double bond is bonded to two different groups. Hence, the given compound exhibits geometric isomerism.
The geometric isomers are cis-isomer and trans-isomer. In cis-isomer, similar or higher priority groups are attached to the carbon on same side and in trans-isomer, similar or higher priority groups are attached to the carbon on opposite sides.
The groups present on either sides of the double bond are given as higher priority groups (
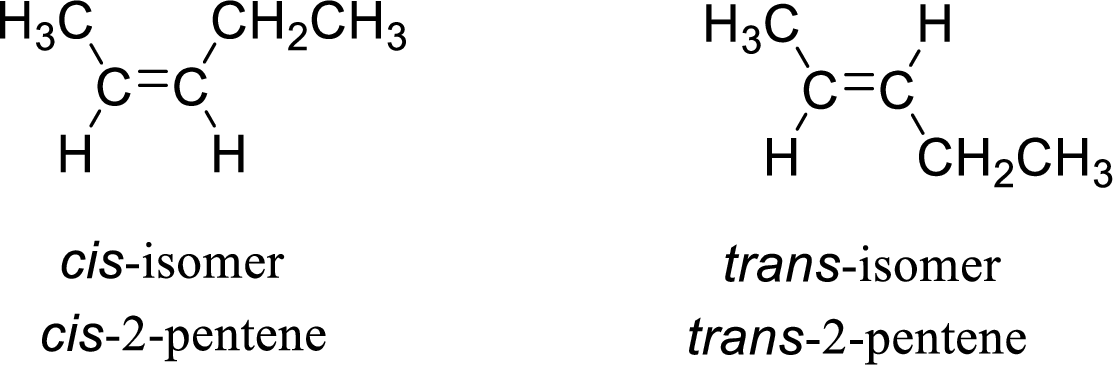
(c)
Interpretation:
The compound 1-chloropropene exhibit geometric isomerism or not has to be given and two isomers has to be drawn and named for the given structure.
Concept Introduction:
The structure of the compound is given by its systematic name.
To give the structure from the name of the compound, the root name has to be identified. The root name indicates the number of carbon atoms present in the longest chain.
Then the functional group (suffix) has to be identified. It indicates whether any functional groups are present in the compound, it also gives whether the compound is an alkane or alkene or alkyne.
The prefix of the name indicates the branched groups and their positions on the carbon chain.
The name of the compound is in the form
Prefix + Root + Suffix
The geometric isomers are said to be the isomers which shows different orientation of groups around a double bond. The geometric isomers are also known as cis-trans isomers.
When two similar or higher priority groups are attached to the carbon on same side, it is said to be cis-isomer.
When two similar or higher priority groups are attached to the carbon on opposite sides, it is said to be trans-isomer.
To exhibit the geometric isomerism, a molecule should have double bonded carbon atoms (
(c)
Explanation of Solution
The given compound is 1-chloropropene.
The propene contains three carbon atoms in chain. As the suffix is –ene, it belongs to alkene group and contains a double bond. The position of the double bond in 1-chloropropene is in between first and second carbon atoms. One chloro group (
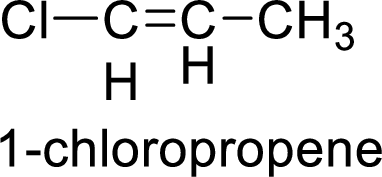
The given compound is 1-chloropropene. The given compound contains a double bond and each carbon in the double bond is bonded to two different groups. Hence, the given compound exhibits geometric isomerism.
The geometric isomers are cis-isomer and trans-isomer. In cis-isomer, similar or higher priority groups are attached to the carbon on same side and in trans-isomer, similar or higher priority groups are attached to the carbon on opposite sides.
The groups present on either sides of the double bond are given as higher priority groups (
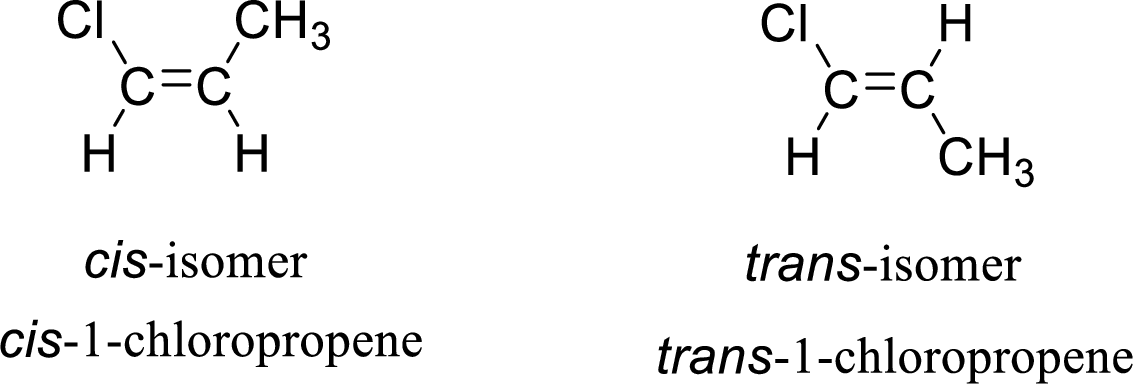
(d)
Interpretation:
The compound 2-chloropropene exhibit geometric isomerism or not has to be given and two isomers has to be drawn and named for the given structure.
Concept Introduction:
The structure of the compound is given by its systematic name.
To give the structure from the name of the compound, the root name has to be identified. The root name indicates the number of carbon atoms present in the longest chain.
Then the functional group (suffix) has to be identified. It indicates whether any functional groups are present in the compound, it also gives whether the compound is an alkane or alkene or alkyne.
The prefix of the name indicates the branched groups and their positions on the carbon chain.
The name of the compound is in the form
Prefix + Root + Suffix
The geometric isomers are said to be the isomers which shows different orientation of groups around a double bond. The geometric isomers are also known as cis-trans isomers.
When two similar or higher priority groups are attached to the carbon on same side, it is said to be cis-isomer.
When two similar or higher priority groups are attached to the carbon on opposite sides, it is said to be trans-isomer.
To exhibit the geometric isomerism, a molecule should have double bonded carbon atoms (
(d)
Explanation of Solution
The given compound is 2-chloropropene.
The propene contains three carbon atoms in chain. As the suffix is –ene, it belongs to alkene group and contains a double bond. The position of the double bond in 2-chloropropene is in between first and second carbon atoms. One chloro group (
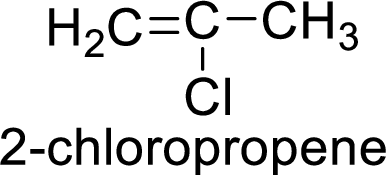
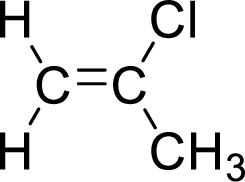
The given compound is 2-chloropropene. The given compound contains a double bond and each carbon in the double bond is bonded to two groups.
The geometric isomers are cis-isomer and trans-isomer. In cis-isomer, similar groups are attached to the carbon on same side and in trans-isomer, similar groups are attached to the carbon on opposite sides.
As 2-chloropropene contains two similar groups (
Want to see more full solutions like this?
Chapter 15 Solutions
GEN CMB CHEM; CNCT+;ALEKS 360
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





