
Concept explainers
Interpretation:
Four isomers consistent with the formula
Concept introduction:
The IHD accounts for the number of rings or multiple bonds the structure could possess.
Number of conjugated C=C double bond are predicted using following table.
| Conjugated |
UV |
Change in |
| - | ||
Answer to Problem 15.32P
Four isomers consistent with the formula
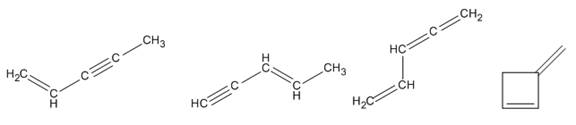
Explanation of Solution
The given molecular formula is
From the table
| Conjugated |
UV |
Change in |
| - | ||
The two conjugated
Therefore four isomers consistent with the formula
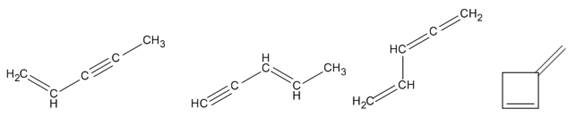
Isomers consistent with the formula
Want to see more full solutions like this?
Chapter 15 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 5. Compare the absorption wavelength of two compounds:1,3-cyclohexadiene and 1,4-cyclohexadiene. Which of the two compounds will show UV absorption at a lower wavelength? What is the possible explanation for that? Include the reference usedarrow_forwardThe structure of the molecule cyclohexene is Does the absorption of ultraviolet light by cyclohexene occur at longer or at shorter wavelengths than the absorption by benzene? Explain.arrow_forwardCalculating the Number of Degrees of Unsaturation in Compounds with O, X, or N Calculate the number of degrees of unsaturation for each molecular formula: (a) C5H8O; (b) C6H11Cl; (c) C8H9N. Propose one possible structure for each compound.arrow_forward
- Chemistry which have multiple UV absorptions which have no UV absorption which has the most intense pi-pi* absorption.arrow_forwardHow do i solve this problem?arrow_forwardChoose an answer An alcohol with molecular formula C4H8O A Ketone with molecular formula C3h6O An alkane with molecular formula C6H14 An akene with molecular formula C5H10 An unsatrated hydrocarbon with molecular formula C6h12 An aldehyde with molecular formula C2H6O A Ketone with molecular formula C2H6Oarrow_forward
- 000 000 Critical absorbance frequency(ies), cm1 Functional group Identity:arrow_forwardHere is the chemical structure of 2-bromobutane: Н Η Н Н HHHH C C- C C-H H :Br Η Н .. Decide whether each molecule in the table below is another molecule of 2-bromobutane, a molecule of an isomer of 2-bromobutane, or a molecule of an entirely different compound. molecule CH₂ CH₂-CH₂-CH-Br CH₂ CH₂ -CH₂ Br HI H H H Η H_ H-C- C-C- H-C-H H H relationship to 2-bromobutane (Choose one) (Choose one) Br: a molecule of an isomer of 2-bromobutane ▼arrow_forwarda) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of spectra)? c) what is different about C=O and O-H bonds compared with C=C or C≡C bonds that show weaker signals?arrow_forward
- Numbering the main chain of branched alkanes For each organic compound in the table below, enter the locant of the highlighted side chain. compound CH₁₂ CH3 - CH, | C - CH₁₂ | - CH2-CH- CH3 CH3 - locant of highlighted side chain ☐ CH3 | CH3 CH2 | CH₁₂ C-CH2-CH CH3 - CH - CH3 ㅁ CH₁₂ CH2 CH3 བos ད ་ CH3-CH2- - C CH3 - CH3 ☐ 00. Ararrow_forward8. Which of the following pairs of compounds is likely to absorb radiation at the longer wavelength and with greater intensity? (a) CH3CH2CO₂H or CH2=CHCO₂H (b) CH3CH=CHCH=CHCH3 or CH3C = C—C = CCH3 OCH3 (c) or CH3arrow_forwardThis is an isomer of C4H10O, and it’s not an alcohol. What could the structure be, and what are the degrees of unsaturation? Which peaks would correspond with which carbons?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

