
Concept explainers
Interpretation:
Four isomers consistent with the formula
Concept introduction:
The IHD accounts for the number of rings or multiple bonds the structure could possess.
Number of conjugated C=C double bond are predicted using following table.
| Conjugated |
UV |
Change in |
| - | ||
Answer to Problem 15.32P
Four isomers consistent with the formula
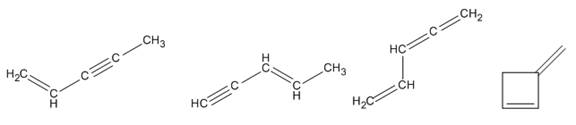
Explanation of Solution
The given molecular formula is
From the table
| Conjugated |
UV |
Change in |
| - | ||
The two conjugated
Therefore four isomers consistent with the formula
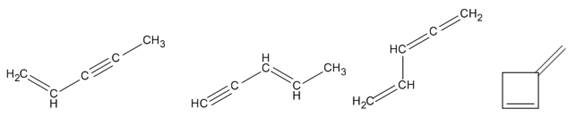
Isomers consistent with the formula
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry: Principles And Mechanisms: Study Guide/solutions Manual (second)
- 5. Compare the absorption wavelength of two compounds:1,3-cyclohexadiene and 1,4-cyclohexadiene. Which of the two compounds will show UV absorption at a lower wavelength? What is the possible explanation for that? Include the reference usedarrow_forwardThe structure of the molecule cyclohexene is Does the absorption of ultraviolet light by cyclohexene occur at longer or at shorter wavelengths than the absorption by benzene? Explain.arrow_forwardCalculating the Number of Degrees of Unsaturation in Compounds with O, X, or N Calculate the number of degrees of unsaturation for each molecular formula: (a) C5H8O; (b) C6H11Cl; (c) C8H9N. Propose one possible structure for each compound.arrow_forward
- Chemistry which have multiple UV absorptions which have no UV absorption which has the most intense pi-pi* absorption.arrow_forwardHow do i solve this problem?arrow_forwardChoose an answer An alcohol with molecular formula C4H8O A Ketone with molecular formula C3h6O An alkane with molecular formula C6H14 An akene with molecular formula C5H10 An unsatrated hydrocarbon with molecular formula C6h12 An aldehyde with molecular formula C2H6O A Ketone with molecular formula C2H6Oarrow_forward
- 000 000 Critical absorbance frequency(ies), cm1 Functional group Identity:arrow_forwardHere is the chemical structure of 2-bromobutane: Н Η Н Н HHHH C C- C C-H H :Br Η Н .. Decide whether each molecule in the table below is another molecule of 2-bromobutane, a molecule of an isomer of 2-bromobutane, or a molecule of an entirely different compound. molecule CH₂ CH₂-CH₂-CH-Br CH₂ CH₂ -CH₂ Br HI H H H Η H_ H-C- C-C- H-C-H H H relationship to 2-bromobutane (Choose one) (Choose one) Br: a molecule of an isomer of 2-bromobutane ▼arrow_forwarda) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of spectra)? c) what is different about C=O and O-H bonds compared with C=C or C≡C bonds that show weaker signals?arrow_forward
- Numbering the main chain of branched alkanes For each organic compound in the table below, enter the locant of the highlighted side chain. compound CH₁₂ CH3 - CH, | C - CH₁₂ | - CH2-CH- CH3 CH3 - locant of highlighted side chain ☐ CH3 | CH3 CH2 | CH₁₂ C-CH2-CH CH3 - CH - CH3 ㅁ CH₁₂ CH2 CH3 བos ད ་ CH3-CH2- - C CH3 - CH3 ☐ 00. Ararrow_forward8. Which of the following pairs of compounds is likely to absorb radiation at the longer wavelength and with greater intensity? (a) CH3CH2CO₂H or CH2=CHCO₂H (b) CH3CH=CHCH=CHCH3 or CH3C = C—C = CCH3 OCH3 (c) or CH3arrow_forwardThis is an isomer of C4H10O, and it’s not an alcohol. What could the structure be, and what are the degrees of unsaturation? Which peaks would correspond with which carbons?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

