
Concept explainers
(a)
Interpretation:
How UV-vis spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction will take place as suggested by change in the
Explanation of Solution
The given reaction is:
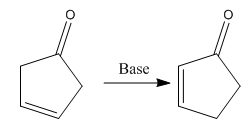
In the above reaction, both the reactant and product have two double bonds; in the product, both double bonds are in conjugation, but in the reactant, they are not.
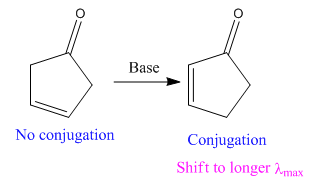
Thus, we can say that the given reaction has actually occurred.
Whether the given reaction has actually taken place is suggested on the basis of conjugation in the product and its effect on
(b)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction does not occur as given, since the
Explanation of Solution
The given reaction is:
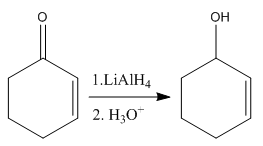
In the above reaction, the reactant has two double bonds in conjugation, and the product has only one double bond. Thus,
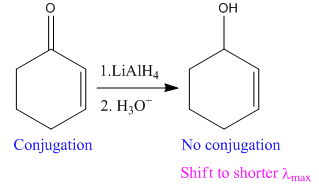
Thus, we can say that the given reaction does not take place.
Whether the given reaction has actually taken place is suggested on the basis of conjugation in the product and its effect on
(c)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction not take place as given, since the
Explanation of Solution
The given reaction is:
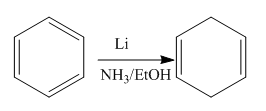
In the above reaction, the reactant has three double bonds in conjugation, and the product has two double bonds not in conjugation. The value of the longest-wavelength
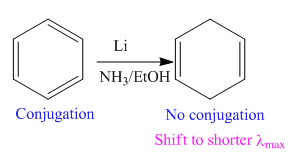
Thus,
Whether the given reaction actually took is suggested on the basis of conjugation in the product and its effect on
(d)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction will take place as suggested by change in the
Explanation of Solution
The given reaction is:
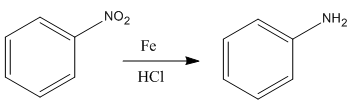
In the above reaction, both reactant and product have three triple bonds in conjugation, but the reactant has a lone pair. So, the reactant’s longest-wavelength absorption corresponds to a
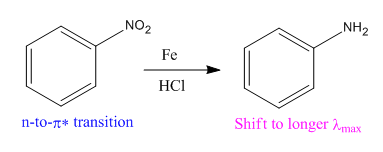
Therefore the
Whether the given reaction actually took is suggested on the basis of conjugation in the product and its effect on
(e)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction actually took is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction will take place as suggested by change in the
Explanation of Solution
The given reaction is:
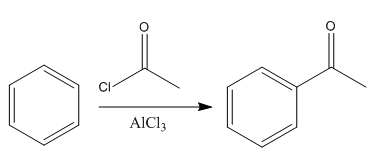
In the above reaction, the reactant has three conjugated bonds, and the product has four conjugated double bonds.
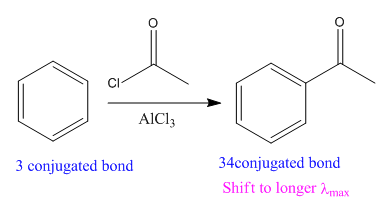
Thus, we can say that the given reaction is actually took place.
Whether the given reaction actually took is suggested on the basis of conjugation in the product and its effect on
Want to see more full solutions like this?
Chapter 15 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- You are doing a ruthenium catalyzed hydrogenation of 4-methylbenzaldehyde (reactant) to 4-methylbenzyl alcohol (product). Draw the reaction equation. Can you use IR spectroscopy to distinguish between reactant and product? Include a detailed list of the characteristic IR bands that you expect to see in your explanation.arrow_forwardHow can you distinguish aldehydes, ketones, and carboxylic acids from each other using IR spectra? Explain using specific examples.arrow_forwardQ8. How would you distinguish the following molecules using IR and Mass Spectrometry? Please label at least two differences for IR and two for MS. Please draw the mechanism for the formation of fragments which can distinguish the two molecules via mass spectrometry. HOarrow_forward
- Can you explain spectroscopy? Let's start with NMR Spectroscopy. What is the difference between a proton being enantiotopic and homotopic? What does it mean for a proton to be shielded or deshielded? How do you determine J (the coupling constant)? What does 'coupling is mutual' mean? How is that translated on the NMS Spectroscopy spectrum? Does that have to do with the polarity of the molecule? Is there an easier way to explain that? When thinking about C-13 NRM Spectroscopy, what it meant by the statement, "the carbons cannot integrate"? When looking at an NMR Spectroscopy spectrum, how do you know which peak belongs to which proton? (The intensity of the peak and the ppm of the peak) P.S. These questions come from after watching a lecture. There is no graded assignment attached to these questions. I simply do not understand the material.arrow_forwardHow can you distinguish between aldehydes and ketones based on Infra-red spectroscopy?arrow_forwardA student has acquired an IR spectra of an unknown six-carbon molecule and has determined that it is either 2-hexyne, 1- hexyne, or 1-hexene. How can the student use their knowledge of IR spectroscopy to determine the identity of the molecule? In your answer, clearly specify what regions of the IR spectrum the student should look at and what bonds are responsible for the absorption bands you indicate.arrow_forward
- in an experiment why would an HNMR be better than an IR spectra?arrow_forwarda) Can these two molecules shown below be distinguished by mass spectrometry? Give reasons for your answer and describe the main fragmentation pathways expected for the left molecule including m/z values of the charged fragments that form. H b) Can these two molecules shown below be distinguished by mass spectrometry? Give reasons for your answer. &arrow_forwardPlease answer this question pertianing to organic chemistry.arrow_forward
- Draw acceptable Lewis structures for the molecular ion (radical cation) formed from the following molecule when bombarded by high-energy electrons in a mass spectrometer.arrow_forward1- While cleaning out a laboratory shelf, you have made a mistake in labeling two bottles of ethanol and acetic acid. To identify the molecular structure of the contents of the bottle, samples were submitted for analysis using infrared spectroscopy, based on the IR results, draw the molecular structure of the molecule that would correspond to the spectrum for that compound. Justify your answer Spectrum A Spectrum B 0. 04 0.2 3000 2000 Waverumber icm-1) 1000 Transmitancearrow_forwardWhat type of spectroscopy would be best to differentiate each pair of molecules? If there is more than one type, please list any/both. Do not include X-ray diffraction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

