
(a)
Interpretation:
The effect of increased pressure over equilibrium for given system should be identified.
Concept Introduction:
Equilibrium: It is where the equal quantity of reactants and products exists as mixture, with the reaction in the forward and backward directions taking place simultaneously.
All
Le Chatelier's principle: When a change is applied to a system at equilibrium, the equilibrium will shift against the change. The equilibrium of a system will be affected by the change in temperature, pressure, and concentration.
Equilibrium Reaction:
Consider a simple equilibrium reaction as follows:
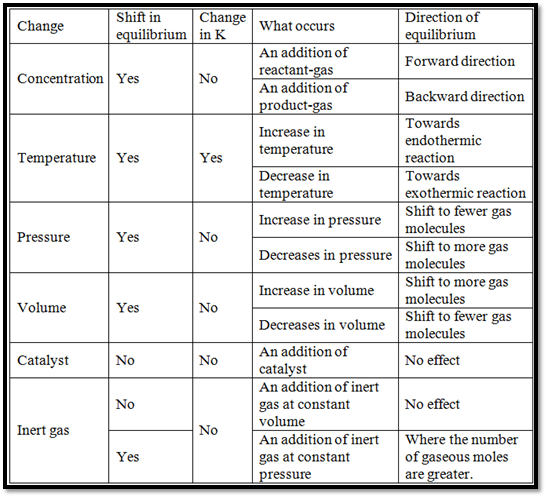
The Factors Affecting Equilibrium are given below:
(b)
Interpretation:
The effect of increased pressure over equilibrium for given system should be identified.
Concept Introduction:
Equilibrium: It is where the equal quantity of reactants and products exists as mixture, with the reaction in the forward and backward directions taking place simultaneously.
All chemical reactions strive to attain an equilibrium between the product side and the reactant side. The actual parameters defining the equilibrium could be the concentration of the respective species or pressure (in case of gaseous species).
Le Chatelier's principle: When a change is applied to a system at equilibrium, the equilibrium will shift against the change. The equilibrium of a system will be affected by the change in temperature, pressure, and concentration.
Equilibrium Reaction:
Consider a simple equilibrium reaction as follows:
The Factors Affecting Equilibrium are given below:

(c)
Interpretation:
The effect of increased pressure over equilibrium for given system should be identified.
Concept Introduction:
Equilibrium: It is where the equal quantity of reactants and products exists as mixture, with the reaction in the forward and backward directions taking place simultaneously.
All chemical reactions strive to attain an equilibrium between the product side and the reactant side. The actual parameters defining the equilibrium could be the concentration of the respective species or pressure (in case of gaseous species).
Le Chatelier's principle: When a change is applied to a system at equilibrium, the equilibrium will shift against the change. The equilibrium of a system will be affected by the change in temperature, pressure, and concentration.
Equilibrium Reaction:
Consider a simple equilibrium reaction as follows:
The Factors Affecting Equilibrium are given below:

(d)
Interpretation:
The effect of increased pressure over equilibrium for given system should be identified.
Concept Introduction:
Equilibrium: It is where the equal quantity of reactants and products exists as mixture, with the reaction in the forward and backward directions taking place simultaneously.
All chemical reactions strive to attain an equilibrium between the product side and the reactant side. The actual parameters defining the equilibrium could be the concentration of the respective species or pressure (in case of gaseous species).
Le Chatelier's principle: When a change is applied to a system at equilibrium, the equilibrium will shift against the change. The equilibrium of a system will be affected by the change in temperature, pressure, and concentration.
Equilibrium Reaction:
Consider a simple equilibrium reaction as follows:
The Factors Affecting Equilibrium are given below:

(e)
Interpretation:
The effect of increased pressure over equilibrium for given system should be identified.
Concept Introduction:
Equilibrium: It is where the equal quantity of reactants and products exists as mixture, with the reaction in the forward and backward directions taking place simultaneously.
All chemical reactions strive to attain an equilibrium between the product side and the reactant side. The actual parameters defining the equilibrium could be the concentration of the respective species or pressure (in case of gaseous species).
Le Chatelier's principle: When a change is applied to a system at equilibrium, the equilibrium will shift against the change. The equilibrium of a system will be affected by the change in temperature, pressure, and concentration.
Equilibrium Reaction:
Consider a simple equilibrium reaction as follows:
The Factors Affecting Equilibrium are given below:

Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Loose Leaf For General Chemistry With Connect Access Card
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





