
Concept explainers
(a)
Interpretation:
Product of hydrolysis of below triacylglycerol in presence of water and sulfuric acid has to be determined.
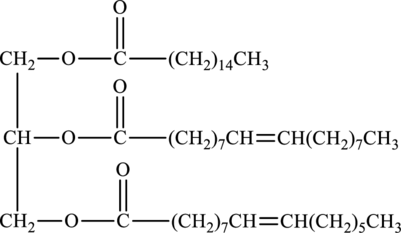
Concept Introduction:
Hydrolysis of ester with water in presence of acid produces carboxylic acid and alcohol. The reaction that explains the hydrolysis of ester is as follows:
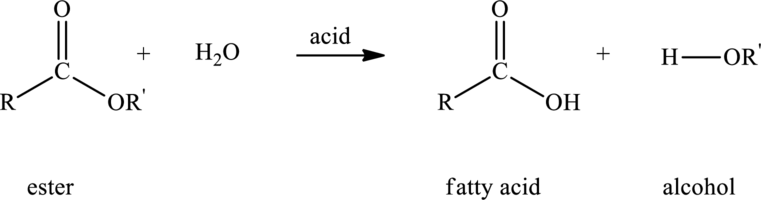
Here,
(b)
Interpretation:
Product of hydrolysis of triacylglycerol in presence of water and sodium hydroxide has to be determined.
Concept Introduction:
Basic hydrolysis of triacylglycerol with water in presence of base produces metal salt of fatty acid and alcohol. The reaction that explains the basic hydrolysis of triacylglycerol is as follows:
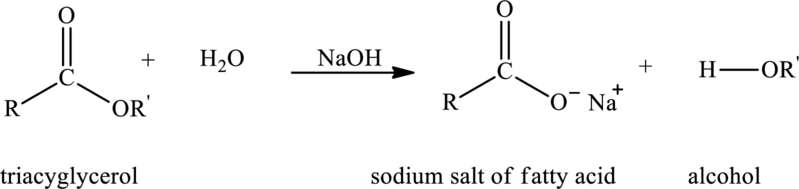
Here,
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Principles of General, Organic, Biological Chemistry
- Draw condensed structural formulas for all products you would obtain from the saponification with KOH of the triacylglycerol in Problem 19-54.arrow_forwardDraw condensed structural formulas for all products you would obtain from the saponification with NaOH of the triacylglycerol in Problem 19-53.arrow_forwardWhy can only unsaturated triacylglycerols undergo hydrogenation?arrow_forward
- 21-100 What are the functions of a cell membrane? To what extent is a bilayer that consists entirely of lipids able to carry out these functions?arrow_forwardThe following is a block diagram for a glycerophospholipid where the building blocks are labeled with letters and the linkages between building blocks are labeled with numbers. a. Which building blocks are fatty acid residues? b. Which building blocks are alcohol residues? c. Which linkages are ester linkages? d. Which linkages involve a phosphate residue?arrow_forwardDraw the condensed structural formula of a triacylglycerol formed from glycerol and three molecules of palmitic acid.arrow_forward
- Draw the condensed structural formula of a triacylglycerol formed from glycerol and three molecules of stearic acid.arrow_forwardA triglyceride can be optically active if it contains two or more different fatty acids.(a) Draw the structure of an optically active triglyceride containing one equivalent of myristic acid and two equivalentsof oleic acidarrow_forward4. Which of the following best describe the physical properties of fatty acids?a. All physical properties of fatty acids are influenced by the length and degreeof unsaturation of their carbon chain.b. Melting points of fatty acids are influenced by the carbon chain length.c. Melting points of fatty acids are influenced by their degree of unsaturation.d. Alltheabove.5. Triacylglycerols are ________.a. insolubleinwater.b. partially soluble in water.c. soluble in water at elevated temperatures.d. solubleinwater.6. The backbone of a triglyceride is __________.a. a three-carbon molecule with an aldehyde and two hydroxyl groups.b. a three-carbon molecule with three hydroxyl groups.c. esterified fatty acids fused with 1 to 3 carbons.d. threelong-chainfattyacids.7. Triacylglycerols are _____ in their ability to store energy when compared to glycogen, the energy storing form of glucose in the human body.a. equally efficientb. insignificantc. less efficientd. more efficient 8. The essential…arrow_forward
- A triglyceride can be optically active if it contains two or more different fatty acids.(a) Draw the structure of an optically active triglyceride containing one equivalent of myristic acid and two equivalentsof oleic acid.(b) Draw the structure of an optically inactive triglyceride with the same fatty acid composition.arrow_forwardThe reaction of glycerol with two fatty acids produces a(n) _____ A) monoglyceride B) diglyceride C) triglyceride D) dilipid E) trilipidarrow_forwardDraw a triacylglycerol with 12 carbon chains that are monounsaturated. Draw the balanced equation for its complete hydrogenationarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning



